In the quest to wring energy from every source imaginable, scientists are putting the squeeze on a common plastic ingredient. Applying force to polymers in water generates enough energy to drive chemical reactions, a team reports online March 1 in Angewandte Chemie.
The technique won’t replace large-scale energy operations, but it offers a way to harness the wisps of unused energy generated by everyday endeavors, like walking or compacting plastic bags at a recycling center. And capturing such energy could lead to cheap, clean ways to sanitize a small container of water, for example, or to run a simple lab bench reaction.
Scientists knew that bonds can break when mechanical force is applied to a polymer. The breakage can generate free radicals, atoms with unpaired electrons that are eager to engage in further reactions. Researchers thought these free radicals hung out wherever they were generated. But the new work shows that when a polymer is squeezed in water, the free radicals migrate and react with the water, generating enough hydrogen peroxide to spur other reactions.
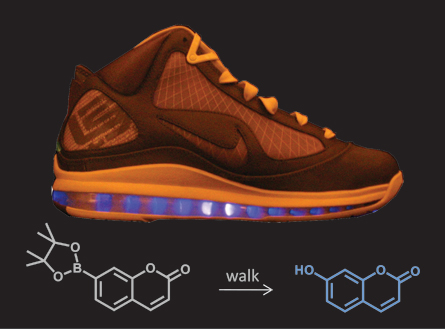
The research team squeezed various polymers, including the plastic PVC and the silicon-based polymer PDMS. When the scientists added gold and silver metal salts to a PDMS tube filled with water, squeezing the tube powered reactions that generated gold and silver nanoparticles. The researchers also injected the sole of a Nike LeBron sneaker with water and a compound that fluoresces when it is cleaved. Half an hour of walking applied force to the polymers in the sneaker’s sole, and the resulting free radicals made enough hydrogen peroxide to cleave the fluorescing compound and make the sole glow.
“People predicted that the energy efficiency would be minute,” says study coauthor Bartosz Grzybowski, a physical chemist at Northwestern University in Evanston, Ill. But he and his colleagues found that capturing and converting the mechanical energy of polymer squeezing into energy for driving reactions can be as efficient as 30 percent. That rate is comparable to some power plants that use coal.
PDMS is used in some medical devices such as catheters, and in some breast implants, raising the issue that normal wear in the body might generate free radicals that could cause inflammation and other problems, notes Grzybowski. But adding an antioxidant such as vitamin E, which latches onto free radicals, to the implant ingredients might be a quick and easy way to mop up the reactive atoms.
Some of the body’s immune cells, though, generate well more hydrogen peroxide than that generated by the polymers, says biomedical engineer Liping Tang of University of Texas at Arlington.
In any case, the fact that enough free radicals are generated to drive reactions — which might even contribute to the breakdown of medical devices — is interesting, Tang says. “I don’t know how unique it is or material-specific, but it could have potential importance.”






