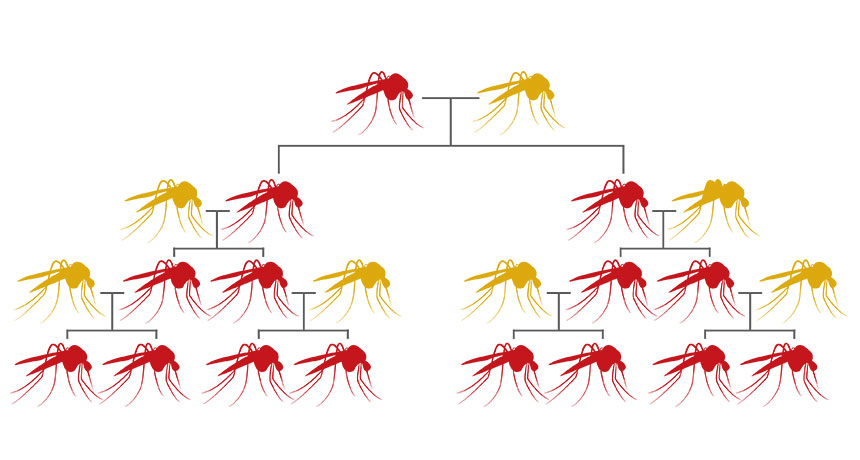
BRAKE AWAY Gene drives cut DNA and paste themselves into an organisms’ genes. This ability enables them to be inherited by a majority of offspring, instead of the 50 percent chance a regular gene has of being passed on to the next generation. Mutations may stop gene drives’ spread, and that might be a good thing, some researchers say.
M. Telfer







