Slime Dwellers
A blanket seeded with microbes appears critical to coral health
Put on your snorkel gear and get close to coral—really close—and you can spy a thin layer of surface slime. Produced continually, and often in prodigious amounts, this mucus can be anything from a thick, soupy liquid to gummy gel. Corals expend significant energy making and replenishing these water-soluble jackets, but scientists have struggled to understand the payoff for this effort.


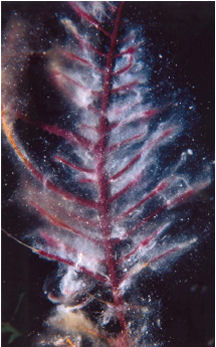

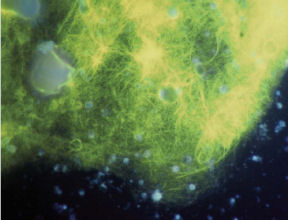
Tiny sea animals that live communally, corals build huge, stony reefs or soft, treelike structures. Within a coral commune, most individuals are clones of their neighbors. Rooted in place, they glean food from the water and periodically eject reproductive cells that drift with the currents before settling at a new site.
For decades, biologists had suspected that the few-millimeter-thick coatings of slime served primarily as protective barriers. The material can prevent corals from drying out when exposed to air at low tide. In some species, coral newborns take sanctuary in the mucus until they graduate to life on their own. And as the slime continually sloughs off, it can carry away sand grains and other debris.
However, researchers are now finding chemical and biological roles for the mucus that rival its barrier functions. Accumulating evidence indicates that most of these activities trace to a large and variable supporting cast of microbes that lives in the mucus. Indeed, some recent studies suggest that certain bacteria can turn surface slime into a germ-fighting medicine cabinet for corals.
Such microbial pharmacists don’t appear to be accidental interlopers. Kim B. Ritchie of the Mote Marine Laboratory in Sarasota, Fla., suspects that at least some antibiotic-making microbes are symbionts—biota that not only aid the coral in fighting pathogenic fungi and bacteria but also derive benefits from living on it.
Corals appear to craft their slimy coatings at least in part to encourage visits by particular families of microbial guests. Ecologists have even begun referring to the welcomed hordes of bacteria, viruses, and more that shelter in the mucus films as integral elements of the coral system.
Like the bacteria that occupy the human gut, the flora that colonizes surface slime can offer health benefits to reef builders. Conversely, an absence of these microbes, caused by stress or environmental change, could leave corals vulnerable to disease.
Mucus microbes may even help corals adapt to new conditions—infections or global warming, for instance—says Eugene Rosenberg of Tel Aviv University. If that’s true, he argues, genes of mucus-dwelling microbes may be as important to coral survival in a period of rapid environmental change as are the genes of the reef builders themselves.
Mucus menus
Recognition that coral health may depend heavily on other species is hardly new. Marine biologists have known for more than a century that a class of single-celled algae called zooxanthellae takes up residence in the majority of healthy corals. Besides imparting color to the otherwise drab reef builders, the microalgae use the energy in sunlight to build inorganic chemicals from seawater into carbon-bearing coral nutrients, such as sugars and amino acids.
Corals also fashion their mucus from these algae-derived ingredients. In fact, “up to 50 percent of the carbon fixed by zooxanthellae is eventually released by the coral as mucus,” notes Christian Wild of Ludwig-Maximilians University in Munich.
Although the mucus mostly consists of sugars and other carbohydrates, the slime may also contain fatty substances, proteins, nucleic acids, and other organic materials. This sticky mix can collect nutrients from the water to augment a coral’s diet, Wild notes. Although corals exhibit rapid growth and metabolism, they typically live in nutrient-depleted patches of ocean. These animals need a means to collect and concentrate nutrients from seawater, and then to keep them from washing away, Wild says. Mucus fills the bill.
Being nutritious, mucus also provides a smorgasbord for microbes that drop by, notes Garriet W. Smith of the University of South Carolina in Aiken. Not surprisingly, microbes in mucus can outnumber those in the same volume of adjacent seawater by 1,000-fold.
Smith’s team has isolated bacteria from coral mucus and examined their dietary preferences by offering the bugs 95 different chemical food sources. Because microbes are fairly selective feeders, these data “give us insights into what’s available as [bacterial food] within the mucus,” he says.
This dietary profiling identified collections of coral microbes that work cooperatively. One group of bacteria will come in and digest chemically complex materials, he explains. By-products of that digestion then become fodder for others.
It’s probably typical for members of a coral species to fashion mucus according to a general recipe. This would explain, Smith says, studies by his group and others suggesting that, regardless of where it lives, a healthy coral species typically harbors many of the same microbes. Microbes won’t dine on a coral’s mucus unless they like its menu, he says, but where that menu appeals, bacteria will come in droves.
Regional cuisines
Although mucus recipes tend to be broadly similar, they can also differ by region and season.
For instance, John C. Bythell and Reia Guppy of the University of Newcastle in England sampled bacteria from the coral Montastraea faveolata at five sites in Caribbean waters off Tobago.
Although all sampled corals harbored many of the same bacteria, the sites also showed slight differences, Bythell notes. Moreover, mucus-dwelling bacteria “were significantly different” than the dominant bacteria in the surrounding seawater, he and Guppy reported in the Dec. 20, 2006 Marine Ecology Progress Series.
During his team’s 8-month study, the mucus microbes on corals at each site underwent substantial changes. One broadly consistent group resided in mucus during Tobago’s wet summer, another during the cooler, drier winter. This suggests that environmental factors affect a coral’s microbial recruits, says Bythell.
Both the seasonal and the site-to-site variations in microbial populations likely trace to alterations in menus offered by a coral’s mucus, he says, although he adds that no one has yet tracked slime composition over time and correlated that information with a coral’s bacterial visitors.
Smith’s group also witnessed a microbial evolution in soft corals known as purple sea fans, Gorgonia ventalina, growing off the Florida Keys. During a 2-year study, the researchers found that most changes in a slime’s microbes were associated with infections of the coral by aspergillus, a fungus.
In the March 23, 2006 Diseases of Aquatic Organisms, the researchers report finding that healthy sea fans hosted a generally consistent mix of bacteria. But, once any part of a fan developed a fungal infection, the microbe population throughout the coral’s mucus—even on tissues free of disease—shifted dramatically. This suggests, Smith says, that the recipe for mucus made by even partially infected corals changes throughout.
Why host bugs?
Although no one disputes that coral slime is rich in bacteria, scientists have long wondered why corals go to the trouble of feeding these hordes. One evolving explanation is that the microbial entourage offers medicinal benefits.
Although they don’t make antibodies, corals demonstrate a primitive ability to fight infection. Going back decades, studies have found germ-killing antibiotics in association with many of these animals.
Such data came mostly from studies in which biologists ground up chunks of coral and sampled all extractable compounds, notes coral ecologist Ariel Kushmaro of Ben Gurion University of the Negev in Beer-Sheva, Israel. As a result, no one knew where the antibiotics came from. Until last fall, that is.
Ritchie’s study of elkhorn corals (Acropora palmata) from reefs in the Florida Keys, published in the Sept. 20, 2006 Marine Ecology Progress Series, was the first to unambiguously tie these antibiotics to mucus microbes, Kushmaro says.
Ritchie collected slime from the corals and isolated their resident bacteria. Previous studies had established that only a fraction of such bacteria can be grown in the lab. Ritchie grew whatever ones she could on sterilized coral mucus, then added pathogens and measured the residents’ survival.
She found very different communities of bacteria—and equally dramatic differences in the ability of germs to coexist with them—during the comfortable spring and the blisteringly hot summer.
For instance, microbe-hosting slime collected in spring from healthy corals inhibited the growth of marine germs or surrogate invaders to one-tenth the growth that occurred with mucus hosting no resident bacteria. Ritchie’s analyses showed that one in five resident-bacteria species exhibited antibiotic activity against a species of bacteria implicated in white pox disease in coral.
During the hot summer, when temperatures rose enough to stress and bleach the corals, the antibiotic activity of the coral slime disappeared. Ritchie attributes this loss to changes in the consortium of microbes inhabiting the slime of stressed corals. Indeed, she showed that when bleaching began to set in, antibiotic-producing guests fled even still-healthy portions of a coral.
Kushmaro and his colleagues similarly find antibiotic activity in at least one-quarter of the microbes cultured from 10 healthy coral species growing in the Red Sea. They presented their preliminary data at an International Symposium on Microbial Ecology last August in Vienna.
A vastly more numerous class of mucus microbes—viruses known as bacteria-eating phages (SN: 7/12/03, p. 26)—may also play an important role in coral health. Focusing on the coral Porites compressa in Hawaiian waters, Rebecca Vega Thurber of San Diego State University has been comparing phages in its mucus with those in adjacent seawater.
Not only are the antibacterial phages far more numerous in the slime, the marine biologist notes, but they also are more likely to be “phages that infect known [coral] pathogens, such as Vibrio harveyi.” Thurber told Science News that when the corals became stressed, “I saw a huge increase in the phage that infects V. harveyi. This tells us that maybe we’ve also gotten an increase in that [bacterial] pathogen, during that stress.”
Encouraging picture?
Although evidence is accumulating that microbes help corals, Rosenberg, Thurber, Smith, and others caution that the link, though tantalizing, remains unconfirmed. It’s impossible to prove how much help bacteria or other microbes provide without comparing corals whose mucus lacks ostensibly beneficial bugs with those whose mucus has been deliberately inoculated with such microbes.
Rosenberg’s group is about to do just that. It’s a necessary test for the “probiotic hypothesis” that his team announced in the December 2006 Environmental Microbiology. Rosenberg, Smith, and other members of the United Nations’ Coral Disease Working Group further describe the idea in the March Oceanography.
Probiotics typically are bacteria-laden dietary supplements for people (SN: 2/3/01, p. 68) or animals (SN: 3/28/98, p. 196). By seeding the body with bacteria that enhance immunity or other beneficial biological functions, probiotic treatments seek to prevent disease or restore health. Lately, marine biologists have begun embracing the idea that corals may exude compounds to lure beneficial microbes into their mucus to create a probiotic community.
The idea makes intuitive sense, Bythell says. Many probiotics target the gut, attempting to overwhelm pathogenic bacteria with a huge, replenishing dose of beneficial ones. Coral mucus resembles mucus lining the human gut. In fact, he says that a medical colleague to whom he showed a photo of a coral’s mucus-producing cells was stunned. “If you told me this was a human gut, I’d have believed you,” Bythell recalls him saying.
Gut flora—largely beneficial bacteria—helps digest foods, boost immunity, and prevent colonization by germs. Corals, one of the animal kingdom’s most primitive multicellular families, appear to accomplish the same thing, he says, using “a mucosal system that’s virtually identical, structurally, to our gut.”
Rosenberg suspects that the symbiosis that develops between various microbes and corals might even have evolutionary significance. For instance, in 1995, Kushmaro linked devastating bleaching incidents in the Mediterranean Sea’s Oculina patagonica coral to an infection by Vibrio shilonii. It was the first time that coral bleaching had been traced to a bacterial infection, and bleaching infections recurred every summer through 2002.
By the following summer, however, the coral’s vulnerability had disappeared. Sampling its mucus turned up no V. shilonii, and deliberately inoculating the mucus with the bug “does not result in coral bleaching,” Rosenberg’s group reports in the May Nature Reviews Microbiology. Indeed, they find that within 4 days, the added germs can no longer be found.
It appears, Rosenberg says, that the corals recruited mucus microbes to quash the would-be killer germs. Certainly, the corals didn’t have enough time to evolve resistance on their own, he says. But by developing new symbiotic relationships with bacteria or phages, they adapted to the germs’ presence.
Similarly, photosynthetic bacteria have been found in some coral tissues, suggesting a way in which the animals might gain sustenance when high temperatures send their algal symbionts fleeing.
Taken together, the new findings give some reason for optimism about the globe’s besieged corals. In view of rising concerns about global warming’s threat to corals, Rosenberg suggests, it could be that corals can adapt to environmental change more rapidly than their own genes would permit.







