Whether bulbous, Roman, or pug, the nose gets all the credit. But the actual star of smell is an unassuming patch of tissue, several centimeters square, tucked up inside each nasal cavity. After a whiff of a peach or a lilac, this tissue captures the volatile chemicals traveling into each nostril. The chemicals bind to receptors on the tissue’s millions of neurons, the neurons relay the information to the brain, and voilà—you know that the peach is ripe and the lilacs are in bloom.


People can recognize some 10,000 different scents. But instead of having 10,000 different receptors, the tissue called the olfactory epithelium does the job with roughly 1,000. Each receptor binds to more than one odor molecule, while each odor molecule binds to more than one receptor. “It’s the overall pattern of the response of all the receptors that the brain interprets as a smell,” says chemist Kenneth S. Suslick of the University of Illinois at Urbana-Champaign.
Since the early 1980s, scientists have used mammalian olfaction as a model to develop chemical-sensing systems. More recently, their efforts have widened to systems that mimic the sense of taste, which also relies on a patterned response to specific chemicals—but in a solution rather than in the air. By creating an array of sensors that offer different kinds and degrees of binding, “one can use a relatively limited number of sensors, combined with the power of pattern recognition, to identify many more substances” than one could do with a set of sensors that each bind to just one target, says chemist David R. Walt of Tufts University in Boston. “If you have enough diversity in the responses, you have the potential to recognize from 10 sensors thousands of different patterns.”
The latest generation of artificial olfactory and gustatory systems uses a variety of binding interactions to produce optical cues, such as changes in color or fluorescence, some of which can be seen with the naked eye. One of the new systems can even discriminate between molecules that have mirror image structures.
The potential applications for the sensing systems range from detecting trace amounts of harmful chemicals to discerning patterns of disease to confirming the quality of your favorite beverage.
But as with noses, “these systems are not going to be useful for doing things like taking a perfume and identifying the different components,” says Walt.
Smell test
Artificial-sensing systems are generally made up of a sensing array hooked to a computer. Just as the brain learns to recognize smells throughout a person’s life, scientists can teach a computer to match patterns on the array to specific molecules or odors. The systems can recognize a complex mixture, such as wine, but they can’t pick out the many chemicals that might contribute to its aroma, says Nathan S. Lewis of the California Institute of Technology in Pasadena.
Scientists called earlier generations of artificial-olfaction systems “electronic” noses because those systems produce electric signals in response to specific chemicals (SN: 2/19/00, p. 125: Available to subscribers at Eau, Brother!).
The system devised by Lewis and his colleagues features a silicon chip overlaid with a checkerboard of polymer films studded with particles of gold, silver, or carbon. The researchers space the particles close enough together so that current can flow among them, says Lewis.
The chemistry of each film determines whether a chemical diffuses into it and makes it increase in volume. As the films swell, they push the metal or carbon particles farther apart, thereby changing the flow of current. A computer detects these changes, uses statistical methods to create a unique pattern, and matches it to a given scent.
The researchers have made chips that detect the nerve-agent analog DMMP, for example, at concentrations of 10 parts per billion (ppb), even in a background of diesel fuel or humidity.
To adapt their sensor to detect volatile amines, such as the irritant butylamine, they included a charged molecule that increased sensitivity to those amines by a factor of 1,000. Lewis’ team describe those sensors in the May 31, 2005 Chemistry of Materials.
Seeing is believing
Two teams are taking a colorful approach to scent sensing.
Suslick and his colleagues have developed an artificial-olfaction system that takes advantage of the diversity of its sensor’s potential chemical interactions. The group incorporates into its system a variety of chemically responsive dyes that can react with a full spectrum of intermolecular interactions. These include hydrogen bonding, acid/base interactions, and metal ion coordination.
A whiff of odor changes the dyes’ colors when a chemical interaction occurs. The researchers image the dyes with a digital camera and then subtract the color present before exposure. “It’s that difference map that’s the fingerprint for the odorant,” says Suslick.
Suslick and his colleagues developed a 36-dye array printed on a square membrane, 1.5 by 1.5 centimeters. The group tested its array against 100 different volatile organic compounds, including amines, alcohols, carboxylic acids, and thiols. The color changes are so distinct, notes Suslick, that the team could identify many of the compounds by sight, even before performing a statistical analysis of the digital image.
The arrays distinguished between even closely related compounds, and they correctly identified compounds more than 99 percent of the time, the team reports in the June 1 Analytical Chemistry.
Because of the many different chemical interactions possible with each dye, “we can tell very subtle differences” between samples, says Suslick.
Walt and his colleagues are also using dyes to visualize the smells picked up by their artificial-olfaction system. They took an optical-fiber array made of tens of thousands of individual glass fibers fused together and inserted a sensing bead into a well etched into the end of each fiber.
The sensing beads are made of either polymer or glass and sport fluorescent-dye molecules. Light enters the fiber array from the non-etched end, travels to the sensing end, and excites the dyes. The emitted light heads back along the fiber to an instrument that gathers the light and creates a colorful image for analysis.
When the scientists expose the system to certain vapors, some chemicals bind to the dyes or diffuse into the polymer. These interactions change the dye’s color, intensity, or both, explains Walt.
The researchers have used a fiber array containing 2,500 beads of six types to detect the explosive chemical 1,3 dinitrobenzene and 3 varieties of coffee beans.
The advantage to using beads is that billions of the same type can be made at a time, says Walt. This way, when beads lose their sensitivity, they can be replaced with identical beads, and there is no need to retrain the computer.
“The cells in our olfactory system are replaced every 6 months,” he says, “but they are replaced by cells that are identical.”
Take a taste
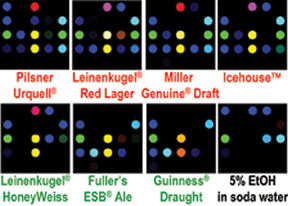
Like smell, the sense of taste and artificial systems modeled after it rely on a patterned response of receptors to certain chemicals. For example, in the July 12 Journal of Agricultural and Food Chemistry, Suslick’s group reports using a 25-dye array to distinguish among 18 commercial beers. The researchers predict that the array may someday indicate whether a particular brand of beer has lost carbonation or become contaminated before going to market.
Eric V. Anslyn of the University of Texas at Austin and his colleagues have reported an artificial system that mimics the way that mammalian taste buds differentiate between amino acids that are mirror images of each other, or chiral. Nature produces a left-handed version of the amino acids, which tend to be flavorless, but chemists have synthesized right-handed versions, which taste sweet, he says.
To detect these amino acids, Anslyn’s team filled each of 96 wells on a plate with a different solution of receptor compounds, all of which were either left- or right-handed, and indicator dyes. Initially, the dyes bound to the receptor compounds. When the scientists added amino acids, they displaced some of the indicator dyes from the receptors, and the dyes changed color. The pattern of displaced dyes identified the amino acid.
The system could distinguish both the type of amino acid and its chiral form, the group reports in the May 3 Journal of the American Chemical Society.
John T. McDevitt of the University of Texas at Austin says that he and his colleagues plan to use an artificial gustatory system to look for “the taste of heart disease,” the pattern of a dozen or so substances in a blood sample that might indicate the health of the heart.
Their sensing system is a silicon square, 1 cm by 1 cm, with a pattern of open, conical baskets that hold sensing beads covered with various chemical receptors. To get a reading, McDevitt’s group first exposes the chip to color-based indicator dyes, then adds the solution to be tested. As in Anslyn’s experiments, the test chemicals bind to the receptors, displacing the indicator molecules and causing the dyes to change color.
To determine a fingerprint for heart disease, the researchers intend to use their chip to simultaneously measure concentrations of inflammation markers and a variety of other proteins in blood that seem to increase risk.
“Heart disease is not one process,” says McDevitt. “It’s many circulatory and cellular issues and clotting factors …. If you focus on just one thing, you miss the problem.”
The researchers have begun by designing a chip that detects C-reactive protein and interleukin-6 in samples of human blood from which the cells have been removed. Unlike sensor systems in which receptors can bind to a variety of chemicals, this application requires one-on-one binding: for example, antibodies that grab specific proteins.
By comparing the blood fingerprints of patients with heart disease with those of healthy people, the researchers expect to find patterns that could “begin to improve risk assessment,” McDevitt says.
No to the nose
Artificial sniffers and tasters shine when a single substance is under scrutiny or the overall fingerprint of an odor or flavor is what’s needed. But when chemists want to pick apart and quantify the various components of a complex mixture, a combination of gas chromatography and mass spectrometry remains the gold standard, says Suslick. Together, these instruments separate a mixture’s components, ionize them, and determine their molecular weight.
There are also situations when a quick yes-or-no response may be most helpful, such as identifying nerve agents, says Walt. “An artificial system is certainly capable of detecting certain nerve agents, but there are multiple steps,” he says, such as the generation of the pattern and computer recognition.
Walt and his team adapted their array system to become a chemical “switch” in response to the nerve-agent analog diethyl chlorophosphate (DCP). After exposure, the beads undergo an irreversible reaction that increases their fluorescence. The arrays light up in less than a second after encountering 13 parts per million vapors of DCP, but they don’t respond to other nerve-agent analogs or mustard gas. The team reports the work in the April 19 Journal of the American Chemical Society.
“You don’t have to wait to do any processing of your signal,” says Walt. “If that thing lights up, you’d turn on the alarm and say ‘Evacuate!'”






