Just as a pepperoni pizza with spicy sausage can leave a person reaching for antacids, a diet high in carbon dioxide can disrupt the delicate balance of the ocean’s chemistry. This odorless, tasteless, invisible gas in Earth’s atmosphere is the ocean’s equivalent of tomato sauce. As the atmosphere mixes with surface seawater, carbon dioxide dissolves into the ocean. Some of the gas reacts with water, forming carbonic acid, H2CO3, the same weak acid that appears in carbonated drinks. The more carbon dioxide in the atmosphere, the more the gas enters into the surface waters.

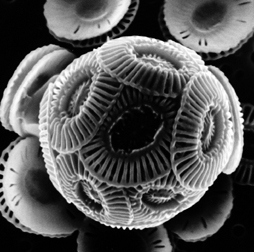
The oceans don’t swing back and forth in acidity with each fluctuation in atmospheric carbon dioxide because they contain a steadying force. It’s their ever-present calcium carbonate, CaCO3–not in mint or berry flavor, but as shells and skeletons of microscopic sea creatures and their dissolved products. The sea floor is lined with this mineral detritus of long-dead organisms, says David Anderson of the University of Colorado in Boulder.
Whether in the ocean or stomach, calcium carbonate works as it might in a high school laboratory experiment in acid-base chemistry, says Henry Elderfield of the University of Cambridge in England. In water, calcium carbonate can dissolve into calcium and carbonate (CO32–) ions and act like a buffer. When a little acid is added to the solution, the carbonate ions neutralize it. For this reason, the ocean’s natural pH–a measure of its acidity–is around 8. That’s close to the neutral 7 on a scale of 0 (very acidic) to 14 (very basic).
But buffers have limits. If too much acid is added to a solution, in a high school lab, it’s a learning experience. It will be quite a different story if the ocean’s calcium carbonate buffer becomes overloaded.
If carbon dioxide concentrations in the atmosphere rise too much, changes in ocean chemistry could disrupt the marine food chain, which is founded on minute, calcium carbonate–making sea creatures. Also troubling is the possibility that the oceans may then remove much less carbon dioxide, a so-called greenhouse gas, from the atmosphere than they do now.
Oceans on the edge
What are the limits of the ocean’s natural buffer? In the present era of rising carbon dioxide concentrations in the atmosphere, this question is especially pressing. By estimates from air trapped in ice cores, the concentration of atmospheric carbon dioxide was about 190 parts per million (ppm) during the last ice age about 20,000 years ago and 280 ppm before the industrial revolution. It’s now about 370 ppm and is predicted to reach 700 ppm by 2100.
In efforts to determine just how much carbon dioxide–driven change the ocean can handle, researchers are looking at fossils of tiny sea organisms, called foraminifera, preserved since the last ice age. The scientists are searching for evidence of significant global changes in ocean pH that might indicate the ocean’s buffering system had been outmatched at times in the past.
The results of one new study of foraminifera shells lead to comforting scenarios in which the ocean, over thousands of years, can handle atmospheric carbon dioxide rises without changing their pH. This evidence flies in the face of work done several years ago. Researchers analyzing boron isotopes in shells had concluded in 1995 that the ocean has become more acidic since the last ice age.
The recent research focuses on the preservation of microfossils–the calcium carbonate remnants of microscopic marine organisms. The basic premise is this: As atmospheric carbon dioxide concentrations rise, the ocean becomes more acidic and calcium carbonate shells dissolve more readily. The chemistry behind this effect is somewhat complex. As carbon dioxide enters the ocean, some of it reacts with dissolved carbonate, which reduces the amount of the ion in the water. As the carbonate ion concentration drops, shells dissolve, releasing the ion into the water.
“We wanted to exploit this effect,” says Anderson.
By examining fossilized shells, he and his coworker David Archer of the University of Chicago inferred the ocean concentration of carbonate ion and, from that, the acidity of the ocean. The scientists described their findings in the March 7 Nature.
Anderson and Archer developed a new technique to compare the abundance of complete fossils of 29 foraminifer species found in both modern and glacial sediment samples at different ocean depths in the Atlantic, Pacific, and Indian Oceans. For years, researchers have studied this foraminifera data to determine the temperature of ancient seas because ocean-surface temperature is the single biggest predictor of a fossil’s abundance in a sample, says Anderson. Dissolution is the second-most-important factor, he says.
Now, it turns out that the carbonate-ion concentration may be teased out of the same data. Some “fragile” foraminifera make shells that are as ornate as a chandelier, so they dissolve more quickly than thicker, hardier shells of “husky” species, Anderson says. The researchers found that–despite varying dissolution susceptibilities of different species–the modern and ancient sediments contained similar ratios of complete shells for the various species. From this evidence, the researchers conclude that there’s no evidence of a global change in pH in the deep ocean, where most marine carbon ends up over thousands of years.
According to Anderson, the finding indicates that if carbon dioxide concentrations in the seas rose over the past 20,000 years, as many scientists contend, then the ocean’s calcium carbonate system effectively buffered against globe-spanning pH changes in the deep sea. Although short-term pH fluctuations may have occurred, he notes, the evidence suggests that the ocean could handle long-term carbon dioxide increases.
However, the problem of ocean acidity isn’t nearly that simple. Wallace S. Broecker and Elizabeth Clark of Columbia University’s Lamont-Doherty Earth Observatory in Palisades, N.Y., also recently examined the dissolution of foraminifera, but they ended up with the opposite result.
Instead of looking for the number of preserved organisms in sediments, Broecker and Clark weighed intact shells for two species of foraminifera. The shell masses have decreased since the last ice age, reflecting an increase in acid-driven dissolution, Broecker and Clark reported in the March 27 Geochemistry Geophysics Geosystems.
Broecker notes that Anderson and Archer’s data were based on glacial-age fossils collected long ago. Although their methods may be sound, their sample might be faulty, he suggests.
Elderfield agrees that Anderson and Archer used a data set that some researchers view skeptically. However, their new method for determining the ancient ocean’s pH is “very clever,” he says. Moreover, he notes, assumptions in both studies could have skewed their respective calculations.
For example, there may have been some as-yet-unknown condition that made the rates of dissolution of calcium carbonate shells in glacial ages and now different. A complication that Elderfield and his Cambridge colleague Stephen Barker report in the Aug. 2 Science is that climate and other factors seem to cause foraminifera to grow lighter shells today than in glacial times, he says.
One thing that these results–and those of other recent studies–do imply, Elderfield says, is that scientists don’t fully understand how the ocean handles carbon dioxide. Without this knowledge, he asserts, researchers shouldn’t push for schemes designed to artificially sequester atmospheric carbon dioxide in the sea.
These have included plans for widespread seeding of oceans with nutrients to stimulate organisms’ uptake of carbon dioxide or for pumping excess carbon dioxide to the seafloor (SN: 6/19/99, p. 392: http://www.sciencenews.org/sn_arc99/6_19_99/bob1.htm).
What’s more, Elderfield adds, although studies like those of Anderson and Broecker aim to discern trends in ocean chemistry spanning thousands of years, they don’t address the most pressing question: What’s going to happen in the next 100 years?
The phytoplankton factor
Researchers at the Alfred Wegener Institute for Polar and Marine Research in Bremerhaven, Germany, are studying shorter-term ocean effects of rising atmospheric carbon dioxide concentrations. They’re performing laboratory experiments with microscopic shell-building phytoplankton known as coccolithophores.
Ingrid Zondervan and her colleagues work with two species of coccolithophores that scientists consider to be responsible for making a large portion of the ocean’s calcium carbonate. The German researchers put the phytoplankton in bottles, exposed them to varying concentrations of dissolved carbon dioxide, and then measured the carbon in the organisms. This enabled the team to calculate shell production for particular concentrations of atmospheric carbon dioxide, including those from pre-industrial times and that expected in the year 2100.
Earlier studies had shown that calcium-carbonate-making marine organisms, such as coral and foraminifera, decrease production of the mineral when ocean concentrations of carbonate ion decrease. This makes sense–there’s less carbonate available for making calcium carbonate. Since rising concentrations of carbon dioxide in the air decrease the amount of carbonate ion present in the ocean despite shells’ dissolving, it seemed plausible that organisms would make less calcium carbonate under conditions of high carbon dioxide, says Zondervan.
Indeed, that’s what the researchers have observed. In one way, however, this result was surprising, says Zondervan. The two species of coccolithophores that she and her colleagues studied make support structures of calcium carbonate inside their cells rather than in exterior shells.
“We thought maybe they could cope with the changes in seawater chemistry–that maybe they don’t depend on it,” says Zondervan. “But they did.”
Using their experimental results and similar ones from another research group, Zondervan and her colleagues modeled the ocean-surface conditions for atmospheric carbon dioxide concentrations projected through 2150 by an international group of scientists, the Intergovernmental Panel on Climate Change (IPCC), in a 1995 report.
She and her coworkers determined that as carbon dioxide increases in the atmosphere, the coccolithophores will not only make less shell but also produce less carbon dioxide, a byproduct of the calcification process.
In this way, the organisms exert a negative feedback on rising carbon dioxide, says Zondervan. She and her colleagues from the Alfred Wegener Institute reported their results in the June 2001 Global Biogeochemical Cycles.
Although this effect might seem comforting to those worried about rises in atmospheric carbon dioxide, the coccolithophores’ reduction of carbon dioxide isn’t nearly enough to counteract the carbon dioxide currently being injected into the environment by human activity, notes Zondervan.
More importantly, she points out, this is only “the immediate effect of a decrease in calcification on the ocean chemistry. We have to see whether there are other effects which we don’t know of–and maybe the effect will be reversed again.”
By way of example, she points out that organisms further up the food chain eat coccolithophores. When the coccolithophores contain less calcium carbonate, bigger creatures might consume more of them because they’re tastier or the drop in calcium carbonate might decrease sunlight reflectance. This could let light penetrate to other phytoplankton in deeper water, causing further perturbations in the food chain and the ocean’s carbonate system.
Even as the scientists realize they have only begun to untangle the intricacies of ocean chemistry in general and of the calcium carbonate cycle in particular, Zondervan says one thing is clear: “We are already changing the ocean chemistry, that’s an experiment that’s already going on.”







