Though often forgotten, the placenta has a huge role in baby’s health
Research in mice draws attention to the importance of the often-overlooked organ
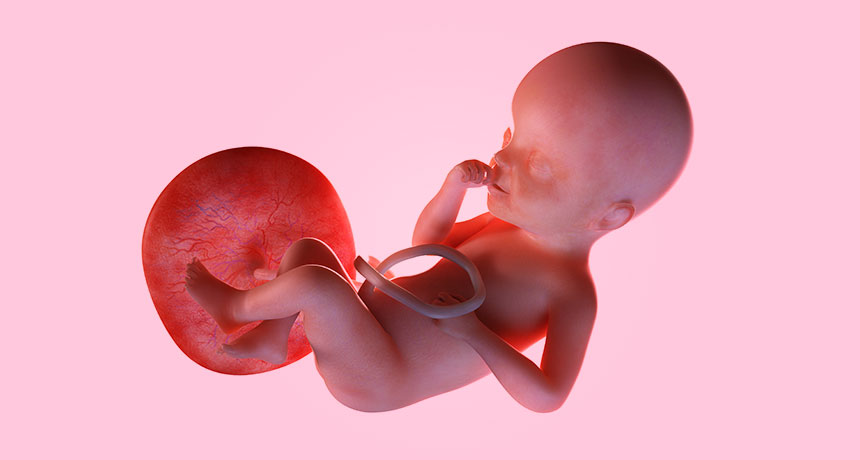
TREE OF LIFE The placenta is a fetus’s lifeline throughout pregnancy. Some problems with embryo development may actually have roots in the placenta, recent research in mice suggests.
Sebastian Kaulitzki/Shutterstock
I am not the first person who has considered composing poetry to the placenta. One writer begins: “Oh Lady Placenta! What a life you lived in magenta.” Another almost coos to the “constant companion, womb pillow friend.” It might sound like odd inspiration for verse, but it’s entirely justified.
This vital organ, which is fully formed by about 12 weeks, nurtures a growing fetus throughout pregnancy, offering oxygen, nutrients and antibodies and eliminating waste. The placental cells forge a deep connection between mom and baby, a symbolic early step in a lifelong bond.
Recent research suggests a placenta that works properly might be even more important than previously thought. Myriam Hemberger of the Babraham Institute and the University of Cambridge, along with colleagues in England and Austria, looked at more than 100 genes in mice that are known to be necessary for an embryo to survive. More than two-thirds of those mouse genes were linked to problems with the placenta. And death of the embryo around days 10 to 15 —when, in mice, the placenta takes over from the yolk sac to supply nutrients — was almost always tied to these placental problems.
The study makes you wonder: How many birth defects in humans might have their roots in the placenta? “You cannot just look at the embryo,” says Susan Fisher of the University of California, San Francisco, who studies how placental cells invade the uterus early in pregnancy. “You should work backward from the placenta.”
Both Hemberger and Fisher believe the placenta is underappreciated. I certainly thought much more about my little embryo turned fetus, growing from sesame seed to grape-sized, grapefruit and beyond, than I did about the disk of tissue supporting my baby-to-be. Fisher calls the placenta “the forgotten organ.”
In 2013, Tina Hesman Saey wrote a feature in Science News about new and growing efforts to understand the placenta. The New York Times published a story the following year, headlined “The Mysterious Tree of a Newborn’s Life.”
Then came the launch of the Human Placenta Project, an initiative of the Eunice Kennedy Shriver National Institute of Child Health and Human Development. Since 2015, the project has invested more than $65 million in the development of technologies that can study the placenta in its natural environment, while a woman is pregnant.
“There is a long history of research involving the placenta, for many, many years,” says neonatal geneticist Diana Bianchi, director of the NICHD, who calls the placenta the Rodney Dangerfield of organs. Like the comedian, it gets no respect. “But the research had pretty much exclusively been after delivery.”
Shortly after a baby is born and his umbilical cord is cut, the placenta is expelled from the mother. At that point, it’s straightforward to inspect its size and blood vessels, and to see if scarring or calcium deposits provide clues to poor function. But it’s much too late to predict a fetus in trouble or to intervene in any way that might ultimately protect mom or baby.
Through the Human Placenta Project, researchers are, for example, using ultrasound to investigate placental blood vessels and blood flow in 3-D during the middle trimester and using magnetic resonance imaging to study oxygen distribution in twins who share a placenta but have separate amniotic sacs. How and when such work might make it to the clinic to affect current and future mommies isn’t yet clear.
Bianchi predicts that a deeper appreciation of the role of the placenta, encouraged through Hemberger’s work and similar studies, could change how pregnant women are monitored in the United States. There’s currently very little monitoring for women in the earliest weeks of pregnancy, the window when the placenta is becoming established. Doctor visits then ramp up as the pregnancy progresses. More attention in the first trimester, including attention to the placenta, could identify women who might be at a high risk for stillbirth or premature delivery, even if they are young and healthy by other standards. “That is a very conceivable, no pun intended, but very likely outcome of this research,” Bianchi says.
After identifying so many genes that affect the placenta, Hemberger’s team went on to show that many of those genes are associated in mice with blood vessel, heart and brain problems in a developing fetus. In other words, problems in the heart may have begun elsewhere, in the placenta. Among three of the mouse genes studied in much more detail, the researchers identified one for which the placenta problem was solely responsible for the death of the embryo at around 10 days. Even if the gene was absent from the embryo, restoring it to the placenta could prolong the embryo’s survival.
If the work holds for humans, it might mean that a fix to the placenta could change the course for a dramatic number of babies who would otherwise be born too small or with birth defects. That’s powerful stuff for would-be moms.
And there’s much more work to be done. “You need to understand normal development before you can address anything that goes wrong,” Hemberger says. A lot of pregnancy complications, including those related to the placenta, originate in the first two to eight weeks, which is still “the black box,” she says.
Recent research out of Japan may help. In January, a team there announced deriving and growing human trophoblast stem cells, the cells that form a large part of the placenta. Though such cells have been studied from mice, this could mark the beginning of more fruitful, detailed efforts to understand placenta formation in people. “Approaches like this,” Hemberger says, “can open up what happens in these early stages.”
Once more is known about how it all begins, it may feel perfectly natural for every person born to sing the praises of the placenta, “The tree of life, the mother-child link, Nourishing baby before it can drink.”
Laura Sanders is away on maternity leave. Elizabeth Quill is Editor, Special Projects for Science News.







