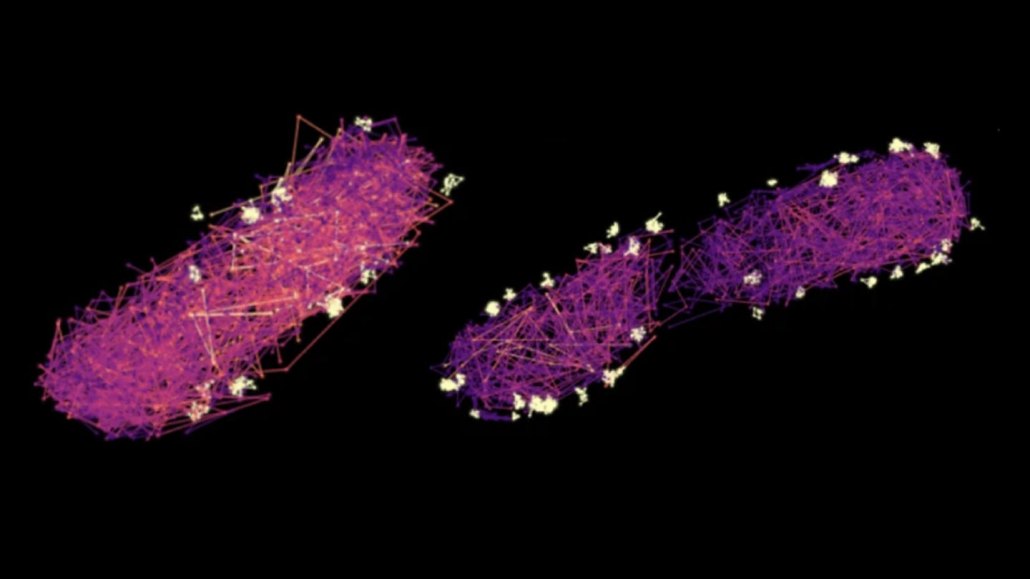
The zigzag paths of proteins that shuttle cargo to the syringe-like secretion systems bacteria use to inject things into their hosts light up in these microscope images (lighter-colored paths correspond to faster movements). In a mutant cell (left) with no cargo to shuttle, the proteins move more quickly than in a normal, secreting cell (right). White blobs mark the drop-off spots.
S. Wimmi et al/Nat. Microbiology 2024