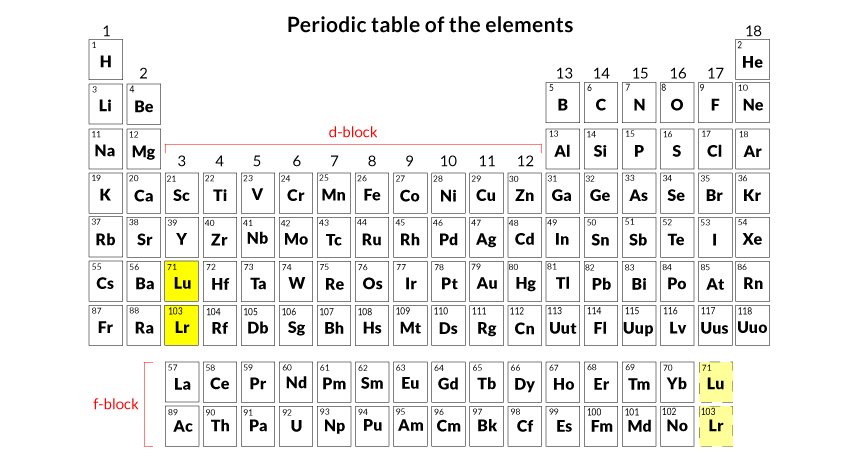
FLEETING Despite new results, controversy remains over where the radioactive element lawrencium (plus its upstairs neighbor lutetium) should be in the periodic table: in the d-block or f-block.
E. Otwell
In their momentary life span, atoms of lawrencium, element 103, may have left a lasting impression on the structure of the periodic table.
For the first time, researchers have measured a basic property of the fleeting radioactive element, namely its ionization potential, they report in the April 9 Nature. The ionization potential — how much energy it takes to strip an atom of its most weakly bound electron —hints at how that atom’s electrons are arranged. Such information for lawrencium may help resolve a long-standing question about where the element belongs in the periodic table.
The question will probably be taken up in the next year or two by the International Union of Pure and Applied Chemistry, says Jan Reedijk, president of IUPAC’s inorganic chemistry division and a molecular inorganic chemist at Leiden University in the Netherlands. IUPAC governs chemical nomenclature. In this case, the union would decide whether lawrencium (along with its relative, lutetium) belongs at the bottom of the periodic table’s third column or at the end of a separate row of elements in an annex below the main table.
Study coauthor Yuichiro Nagame, a chemist at the Japan Atomic Energy Agency in Tokai, didn’t expect the experiment to spark debate about the periodic table. “I’m very happy to learn of these discussions,” he says.
Nagame and colleagues set out six years ago to study lawrencium’s electron structure, a steep technical challenge because typical methods require about a trillion atoms. Lawrencium, named after Ernest Lawrence, the inventor of the cyclotron, is transient and extremely difficult to make. Nagame and colleagues managed to take the measurement by generating about a thousand atoms in total.
By firing a stream of boron ions (element 5) into californium atoms (element 98), Nagame and colleagues created an isotope of lawrencium. The collisions created a new lawrencium atom every few seconds, which had a 27-second half-life. With a new method that sweeps lawrencium up into a helium-based atmosphere, the researchers quickly shot the ephemeral atoms into a metal tube heated to either 2,700 or 2,800 kelvins.
Contact with the hot tube can pluck electrons from lawrencium atoms, creating ions. By sending other elements through the tube beforehand — elements with known ionization potentials — Nagame and colleagues came up with an equation to calculate ionization potential based on how many ions formed in the tube.
After counting how many lawrencium ions tumbled out, the researchers calculated that lawrencium has an ionization potential of about 4.96 electron volts, which is low. In fact, it’s the lowest of all the elements in the appended group of elements at the bottom of the table, where some chemists say lawrencium belongs. The low ionization potential suggests that lawrencium has a peculiar arrangement of electrons.
Heavy elements like lawrencium have a high nuclear charge, which causes electrons near the nucleus to move fast. As Einstein’s theory of relativity predicts, speedy electrons gain mass, causing inner groups of electrons to form dense clouds around the nucleus. This shields the nuclear charge, leaving electrons on the outskirts of the atom’s structure to loosen and rearrange.
Electrons arrange themselves in cloudlike regions around the nucleus called orbitals. These orbitals are designated by numbers for their energy level and letters (s, p, d, f) for their shape. For example, s orbitals form spherical clouds around a nucleus. Based solely on lawrencium’s atomic number, chemists expect that its outermost electrons will be in 5f, 6d and 7s orbitals. But the low ionization potential suggests that lawrencium has one very weakly bound electron in the 7p orbital instead of the 6d.
The empty 6d orbital suggests to some chemists that lawrencium shouldn’t be in the third column of the periodic table. That column sits in a region of the table known as the d-block, which generally includes elements with electrons occupying an outer d orbital. The study confirms that lawrencium belongs in the table’s annex, says chemist Laurence Lavelle of UCLA. That separate group of elements, known as the f-block, generally contains elements with electrons inhabiting an outer f orbital.
Others suggest that, in this case, using orbitals to determine placement in the periodic table is murky. Instead, other trends in the table such as those seen in the actual numerical values of ionization potentials are more helpful. The measured ionization potential supports the idea that lawrencium does “not belong at the end of the f-block but rather at the start of the d-block,” says chemical historian William Jensen, retired from the University of Cincinnati.
One property isn’t enough to make an argument for placement, says chemical historian and philosopher Eric Scerri of UCLA. “We need a more global approach,” he says.
Scerri thinks that the periodic table should be widened to have 32 columns so that the f-block elements can merge into the main table, and all elements would be displayed in order by atomic number. That revision would place lawrencium in the d-block. With the new data, Scerri is working on a report to persuade IUPAC to approve a new table with lawrencium’s d-block position.
Reedijk, of IUPAC, looks forward to Scerri’s report. But he cautions that IUPAC’s deliberations will probably be slow. When IUPAC proposed modifying the periodic table’s column numbering in 1985, it took about five years to decide and another 10 to 15 years for chemists to adopt the changes, Reedijk says.
Editor’s note: This article was updated on April 17, 2015, to specify the number of lawrencium atoms generated. On April 22, the main image was updated.






