No New Meds
With drug firms in retreat, the pipeline for new psychiatric medications dries up

Michael Morgenstern
Psychiatry seemed poised on the edge of a breakthrough. In early 2011, after decades of no radically new drugs, a fundamentally different schizophrenia treatment promised relief from the psychotic hallucinations and delusions plaguing people with the disease. The new compound, devised by chemists at Eli Lilly and Co., hit a target in the brain that older medicines had ignored.
All signs pointed to success. In mice, a similar molecule could block the schizophrenia-like effects of PCP. In people the new drug, LY2140023, appeared to curb psychotic behavior with few side effects, small pilot studies showed. In March 2011, Lilly began enrolling 1,100 people in a definitive Phase III clinical trial, the final test designed to show conclusively that the new compound worked.
A year and a half later, the drug was dead. After years of work and millions of dollars of investment, the failure was crushing. People with schizophrenia were no better on the new drug than similar people taking a placebo, early results indicated. “I’m disappointed in what these results mean for patients with schizophrenia who still are searching for options to treat this terrible illness,” Jan Lundberg, president of Lilly Research Laboratories, said in a press release.
Although the results were devastating, many in the field weren’t surprised. For new drugs designed to treat complex brain disorders such as schizophrenia, depression and anxiety, the odds of success are exceedingly slim. Given the current state of affairs in the drug discovery world, some would argue those odds are close to zero. Not a single drug designed to treat a psychiatric illness in a novel way has reached patients in more than 30 years, argues psychiatrist Christian Fibiger of the University of British Columbia in Kelowna, who described the problem in a 2012 Schizophrenia Bulletin editorial. “For me, the data are in,” says Fibiger, who has developed drugs at several major pharmaceutical companies. “We’ve got to change. This isn’t working.”
Fibiger is not alone in thinking the existing approach needs a radical overhaul. Psychiatrists and neuroscientists around the world recently have begun sounding the alarm that the field is in crisis. Drug development for complex psychiatric illnesses is misguided, they argue, stuck churning out slight variations on therapeutic themes that didn’t work all that well to begin with. Faulty assumptions, animal models that don’t look anything like human diseases, hazy diagnoses and a lack of knowledge about how the brain works have all thwarted the search for better drugs.
Of course, fixing a brain poses challenges that don’t apply to other body parts, says neuroscientist Steven Hyman of the Broad Institute of MIT and Harvard. “You can’t just open up the hood, take out a chunk and see what’s happening,” he says. And even if that were possible, it probably wouldn’t add much clarity, Hyman argues in the October 10 Science Translational Medicine. “Brain research is really hard,” says Hyman. “No one should be blamed for how hard this is. But we did get stuck.”
Pharmaceutical exodus
At a meeting of the American College of Neuropsychopharmacology late last year, this crisis was the predominant theme. “It’s become a topic with a lot of talk and no idea of where to go,” Hyman says.
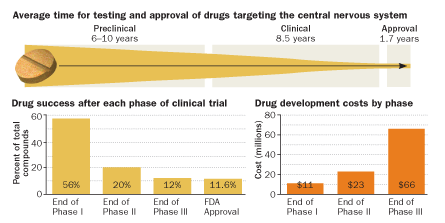
Drug discovery is a tough, slow business. Initial exploratory work to identify a molecular target and a drug that will interact with that target can take years. After that, refined studies are conducted in animals, typically rodents, and then the experiment eventually moves into people. Brain drugs take about 18 years on average to go from preclinical experiments to approval.
This glacial pace is frustrating to the staggering number of people who need better therapies — and to the doctors who treat them.
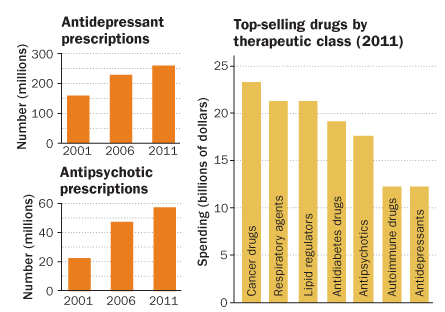
Many current drugs do exist — by one count, more than 50 different psychiatric medicines together garnered $25 billion in sales in the United States in 2011. But these treatments often don’t deliver. “There may be a lot of medicines out there, but they’re not doing what we need them to do,” says Thomas Insel, director of the National Institute of Mental Health.
Pharmacies in the United States, for example, filled about 250 million prescriptions for antidepressants in 2011. But these drugs don’t work in many people and, even if they do, take weeks to kick in. Antipsychotics, for which roughly 55 million prescriptions were filled in 2011, often do nothing for the most serious symptoms of schizophrenia. On top of that, many of these medicines have side effects so objectionable that people stop taking them.
Despite a dire need for better treatments and a large market — one in four Americans suffers from a diagnosable mental illness in any given year — many drug companies are retreating. Though some small, targeted efforts remain in place, pharmaceutical giants GlaxoSmithKline, AstraZeneca and Novartis recently shuttered their main brain drug discovery programs. “It’s pretty scary when you get down to it,” says Kenneth Kaitin, director of the Tufts Center for the Study of Drug Development.
This exodus makes sense: Companies can’t afford to spend so much time and money only to have a drug fail in Phase III trials, as LY2140023 did.
A survey of pharmaceutical and biotechnology companies revealed the perils of investing in drugs that target the brain. These drugs are more likely to fail and leak out of the pipeline than other kinds of medications. And brain-targeting drugs spend an average of 8.5 years in human tests alone, more than two years longer than the average for other kinds of drugs. “These tend to be very difficult, expensive clinical trials,” Kaitin says. Companies that endure a late-stage failure of a drug after years of testing take a huge financial hit. “Very few companies can withstand that,” he says.
Drug firms are also feeling the squeeze from generics, cheaper versions of a drug that can be sold after a certain length of time by companies that didn’t have to pay for the original development and testing. To stay profitable, developers need to come up with a fundamentally new drug. “It’s really a breakthrough or nothing,” Kaitin says. “And breakthroughs are hard to come by.”
Some researchers point to reasons for hope. Biomedical advances such as genetic sequencing and brain-scanning technology may usher in a deeper understanding of these complex disorders. Many experts, though, argue that for these discoveries to translate into help for patients, things have to change.
Playing it safe
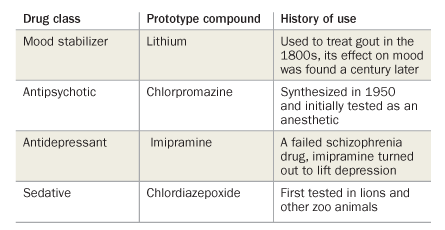
Most psychiatric drugs in use today originated in serendipitous discoveries made many decades ago. In 1952, doctors noted that patients on the antituberculosis drug iproniazid became euphoric. The observation launched iproniazid, the first antidepressant. A version of the schizophrenia drug chlorpromazine was originally tested in the 1950s as an anesthetic. Around that time, a French surgeon recognized the drug’s potential in psychiatry, noting that before surgery patients on the drug became “calm, somewhat somnolent, and relaxed.”
Since then, most new psychiatric drugs have been subtle variations on these and a handful of other original molecules. “You get lucky by finding a medicine that helps,” Insel says. “Then you create another medicine that looks slightly different.”
Derisively termed “me too” drugs, these subtle iterations only rarely make a difference to patients. Instead, Fibiger says, they are created to pad the pockets of drug companies, which aggressively market the drugs as the next new blockbuster. Some are touted as having milder side effects than their predecessors, but critics contend that those side effects are often no better, just different.
As an example, the antipsychotic clozapine, developed in the 1960s, can in rare cases dangerously lower white blood cell counts. Later drugs were developed to mimic clozapine’s activity without lowering white blood counts. But the second-generation drugs lead to substantial weight gain and serious metabolic problems.
This copycat process might be a by-product of looking where the light shines brightest. Even basic researchers have a habit of studying what other people are studying. And without an influx of brand new ideas, drug developers are left spinning their wheels. “Even though the technology got better and better, frankly, our success rate got worse and worse,” Fibiger says.
After the discoveries of chlorpromazine and another antipsychotic drug, haloperidol, in the 1950s, scientists figured out that these drugs changed the brain’s levels of the chemical messenger dopamine. Since then, the relationship between dopamine and schizophrenia has been hotly pursued by the research community, even though it is not exactly clear how those drugs work to combat symptoms. Thousands of studies have been published describing the link between dopamine and schizophrenia. In turn, all of the current drugs for schizophrenia target the brain’s dopamine system. (LY2140023 hit a different pathway in the brain called the glutamate system.)
Dopamine probably does play a role in schizophrenia, but other still unexplored factors might be as or more important. The cause of the disease remains unclear. Studies that focus on these neglected unknowns might offer the insights needed to bring about better, faster and more effective drugs, Hyman says.
Another problem that stymies breakthroughs is a heavy reliance on animal models. Scientists often use mice to look for symptoms that can then be applied to human diseases. A mouse that quickly gives up on trying to swim in a tub of water is thought to be despondent. A mouse that doesn’t sniff as much as normal around a new mouse is said to be antisocial.
Although these animal behaviors are often the best option for study available, they are a far cry from the human diseases they stand in for, Hyman says. So drugs that can fix these problems in mice don’t necessarily translate to people. “Right now, we are in a period of disillusionment with animal models,” he says. “People are tired of curing mice.”
Hyman believes that human stem cell technology might offer a better solution. Ideas — and eventually drugs — might be tested on groups of carefully cultivated human nerve cells in a dish, for instance. Going further, Hyman and others have started talking seriously about small, carefully designed experiments on people (with oversight and consent, of course). In April, a workshop at the Institute of Medicine will explore the idea of testing drugs first in humans.
Brain incognito
Perhaps the largest impediment to the development of new psychiatric drugs is the brain itself. A complex web of interconnected systems constantly altered by the environment, the brain is difficult to study.
Even though it’s nestled right in our heads, the brain is hard to reach. A blood pressure cuff can be slapped on for an instant and objective measure of what’s happening with the heart. A needle biopsy can physically pull out suspected breast cancer cells for further tests. But when it comes to the brain, there is no easy way to identify and measure the thing that isn’t working.
When something goes wrong in the brain, as it does in mental illness, the only outward signs are symptoms. And while these symptoms often signify a particular disease, they are far from perfect indicators. For one thing, a particular symptom can accompany multiple diseases. Trouble sleeping often surfaces in depression, schizophrenia and anxiety disorders, for instance. Unlike an unambiguous blood pressure reading, symptoms are subject to interpretation by both patients and doctors, who often rate symptoms on a sliding scale of severity. To muddy the waters even more, symptoms of mental illness can fluctuate, appearing and disappearing over time. Catching someone on an unusually bad or good day can confound a clear view of the disorder.
And even disease diagnoses, often reached by consulting a list of common symptoms, may not offer much insight into what’s going on in the brain. Different brain pathologies can yield diseases that appear similar, yet have entirely different causes. Making things even more difficult for would-be developers of new psychiatric medications is the fact that on a biological level most mental disorders are not well understood at all.
All of these factors help explain why it has been impossible to develop a psychiatric drug that makes a disease go away in every person who takes it. In a clinical trial of people with depression, for example, treating patients as one homogenous group obscures meaningful results. If a small subset of the people in the study respond to the drug beautifully, for example, but most people don’t, the drug would appear to fail.
“This field is going to have to get past the idea that there will be a perfect pill for these disorders,” Insel says. What’s needed is a deeper understanding of the brain — the genes, the molecules, the circuits that go awry in some diseases, he writes October 10 in Science Translational Medicine. It’s much harder to fix something if you don’t know what’s going wrong.
A reset
The situation is grim, but not hopeless, says Insel. At a time when major pharmaceutical companies are abandoning psychiatric drug development, Insel says he is doubling down, investing federal grant money in places where investors fear to tread. “There are a whole series of pretty amazing developments that I think are worth investing in,” he says.
One such project is a newly created funding opportunity for scientists called the Research Domain Criteria, or RDoC. This project has the audacious goal of mapping particular symptoms or behavioral abnormalities to specific causes in the brain. RDoC will bypass the current onerous and problematic disease labels and instead directly investigate what’s going on in the brain. Rather than attempting to tie the umbrella disease of schizophrenia to a certain kind of neurotransmitter in the brain, under the RDoC plan a specific part of the disease — hearing voices, for example — might be linked to that neurotransmitter.
Doing small, quick, early-stage trials of prospective compounds in people is another way to move more drugs through the pipeline. In many cases now, a failure in a clinical trial is completely uninformative, Insel says, since it’s unclear why a compound failed. By carefully designing studies to test whether a drug hits its target and eases some measurable outcome, these “fast-fail” trials could rapidly identify both promising drugs and ones that don’t work. NIMH has requested grant proposals for fast-fail trials aimed at schizophrenia, autism and mood and anxiety disorders.
Some researchers say that the time has come to get back to the roots of psychiatric drug discovery, in which people were given drugs and observant clinicians paid careful attention to the drugs’ effects. This is the principle behind the upcoming first-in-humans workshop. And it is the kind of careful observation that can liberate a drug, freeing it to treat problems that it wasn’t initially designed to fix. This is how a TB drug and an anesthetic ended up as mood treatments.
Even with many firms pulling back, some pharmaceutical companies are teaming up to work on these tough psychiatric disorders, Kaitin says. Merck and other companies are starting to enter into collaborative agreements with each other and academic centers, spreading the risk but potentially sharing the profits. “I paint a pretty dismal picture when I go out and talk about this, but I think the future is going to be in partnerships and collaborations,” Kaitin says.
And of course, more basic experiments on how the brain works will prove instrumental to designing better drugs. If support for that sort of undirected experimentation dries up, so will drug companies’ efforts to turn those discoveries into medicine. “At some point, you’re going to exhaust the supply,” Kaitin says.
Despite the challenges, people are starting to talk seriously about ways to change how psychiatric drugs get discovered, Insel says. “I’m really optimistic,” he says. “I think there are great opportunities here.”







