
GUT REACTION Research on rats and mice suggests that gut bacteria start the cycle of obesity. The bacteria make a molecule called acetate that sends signals through the vagus nerve to stimulate hunger and fat storage.
stester/Shutterstock
Gut microbes cause obesity by sending messages via the vagus nerve to pack on pounds, new research in rodents suggests.
Bacteria in the intestines produce a molecule called acetate, which works through the brain and nervous system to make rats and mice fat, researchers report in the June 9 Nature.
If the results hold up in humans, scientists would understand one mechanism by which gut microbes induce obesity: First, the microbes convert fats in food to a short-chain fatty acid called acetate. Acetate in the blood somehow makes its way to the brain. The brain sends a signal through the vagus nerve to the pancreas to increase insulin production. Insulin tells fat cells to store more energy. Fat builds up, leading to obesity.
Acetate also increases levels of a hunger hormone called ghrelin, which could lead animals and people to eat even more, says Yale University endocrinologist Gerald Shulman, who led the study.
“This is a tour-de-force paper,” says biochemist Jonathan Schertzer of McMaster University in Hamilton, Canada. Most studies that examine the health effects of intestinal microbes just list which bacteria, viruses, fungi and other microorganisms make up the gut microbiome, Schertzer says. But a catalog of differences between lean and obese individuals doesn’t address what those microbes do, he says. “What’s in name?” he asks. “When you find a factor that actually influences metabolism, that’s important.”
Shulman and colleagues didn’t set out to study the gut microbiome’s role in obesity. In an unrelated study, the researchers noticed that infusing rats with acetate caused the pancreas’s beta-cells to pump out insulin. Feeding rats a high-fat diet also caused acetate levels in the rodents’ blood to increase and stimulate insulin production.
Where the acetate was coming from wasn’t known. Previous studies had shown that gut bacteria can make acetate and other short-chain fatty acids (some of which have been linked to good health). So Shulman and colleagues wondered if gut microbes were responsible for the rodents’ acetate production. In the new study, rats treated with antibiotics to kill off their gut microbes and “germ-free” mice raised in sterile conditions didn’t produce much acetate. Restoring these rodents’ gut bacteria boosted acetate production, especially when the animals ate high-fat diets. Those results and others confirmed that the acetate comes from gut microbes.
Story continues after sidebar
The nerve
Obesity may start with gut microbes, new research in rodents suggests. Gut bacteria turn fat in food into acetate. Acetate then signals the brain to send messages through the vagus nerve to the pancreas and stomach. Beta-cells in the pancreas pump out insulin, which causes fat cells to store more fat. The memo to the stomach tells it to make a hunger hormone called ghrelin, which may induce overeating.
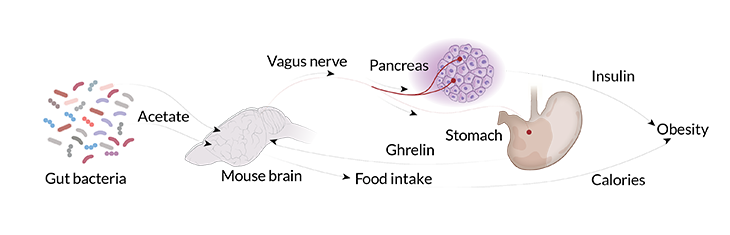
Acetate doesn’t directly stimulate pancreatic beta-cells to make insulin, Shulman and colleagues discovered. Instead, acetate somehow influences the brain to send a “make-insulin” signal through the vagus nerve to the pancreas. The vagus nerve is a major nerve system that connects the brain and internal organs, allowing messages to be passed both ways.
In the study, injecting acetate directly into rats’ brains stimulated insulin secretion. Cutting the vagus nerve or blocking its activity with drugs stopped acetate from boosting insulin levels. Those results indicate that the vagus nerve is carrying the message that prompts fat storage and may be an important player in obesity. The nerve has already been implicated in a variety of other ailments; stimulating it may offer some relief for conditions such as rheumatoid arthritis, heart disease and diabetes (SN: 11/28/15, p. 18).
In January 2015, the U.S. Food and Drug Administration approved a vagus nerve stimulator for treating obesity. That device is supposed to signal the brain that the stomach is full. But Schertzer worries that acetate in the blood could still make it to the brain and override the all-full message. Blocking the signal from the brain to the pancreas may be more effective, he says.
If researchers confirm the same process happens in people, then identifying the bacteria that make acetate and figuring out how to stop them could lead to new obesity treatments, says microbiome researcher Chad Trent. The discovery of this possible obesity mechanism will open new research avenues, predicts Trent, of the New York University Langone Medical Center. “I really admire this paper,” he says. “It could turn out to be foundational work in the field.”
Editor’s note: This story was updated June 17, 2016, to clarify when the FDA approved a vagus nerve stimulator for the treatment of obesity.






