Chemical engineers have devised a simple way to create better polymer-based coatings for products ranging from surgical implants to ship hulls.
The researchers have already used the method to make water-resistant coatings that last much longer than ones prepared by more conventional means.
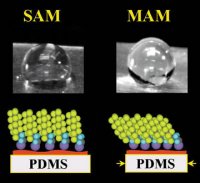
Materials designers often rely on chemistry to modify a surface. Using a conventional method, researchers choose a material—such as a light, flexible polymer—and then add small, chainlike molecules that line up on its surface. Each of these molecules has one end that sticks to the polymer and another end that bestows the resulting material with some desirable surface trait, such as water resistance.
This method has a significant limitation, however. The deposited molecules arrange themselves no more tightly than nature permits them to pack on their own. Water squeezes between the relatively loosely aligned molecules and quickly degrades the polymer.
Now, Jan Genzer and Kirill Efimenko of North Carolina State University in Raleigh have found a way to jam self-assembling surface molecules into denser, more stable arrangements. They reported the work last week in Honolulu at the 2000 International Chemical Congress of Pacific Basin Societies and in the Dec. 15 Science.
The researchers first stretched a film of a common elastic polymer called poly(dimethyl siloxane), or PDMS, which is used for caulking, lubrication, and various other purposes. They let molecules with water-resistant tails line up on the stretched surface. As expected, the molecules’ heads chemically stuck to the surface and their tails pointed away. The scientists then released the tension in the polymer, which sprang back to its original area. As a result, the surface molecules drew more tightly together.
“We were able to fool Mother Nature,” says Genzer. Tests indicated that the new material, with its unnaturally dense surface, retained water resistance throughout a 7-day, underwater trial and for 6 months while sitting on a humid shelf. Coatings prepared without the new stretch-and-release step kept water at bay for only a day when submerged, adds Genzer.
“I think this is a very significant finding,” comments Manoj K. Chaudhury of Lehigh University in Bethlehem, Pa. “The idea is very simple. However, the results are spectacular.”






