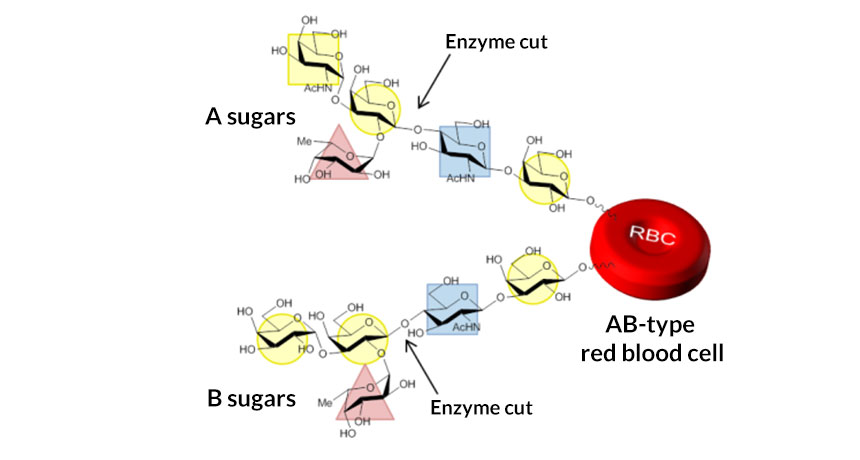
TYPE ALL With engineering, a natural enzyme can better cut off some sugars that determine if a blood cell is type A, B or AB (as shown), resulting in a generic blood type that is safe for all people.
D.H. Kwan et al/Journal of the American Chemical Society 2015







