Television viewers may be tossing out their old rabbit ears in favor of sleeker digital receivers, but scientists are raising the microscopic equivalents of antennas to new prominence.
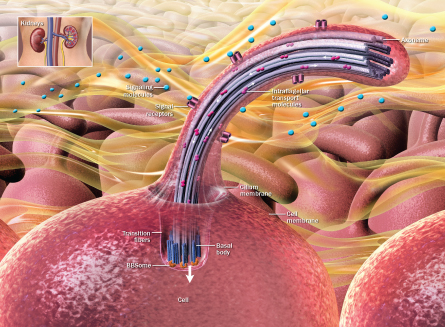

Most cells in the body, from light-gathering eye cells to kidney cells to brain cells, sport a single, prominent hairlike structure sticking out like an index finger flashing the No. 1 sign. While cells can have other protrusions that serve as propellers or sweep away mucus and debris, the No. 1 “primary” cilia don’t whip or wiggle or brush anything along. For a long time, in fact, scientists have thought about primary cilia the way people think about their appendixes, as vestigial organs that may once have had a purpose but are largely useless today.
Evidence now suggests that primary cilia aren’t just stray whiskers evolution hasn’t gotten around to shaving off. Instead, these structures might be among the most important that a cell possesses. Scientists are coming to see primary cilia as a major means by which a cell communicates with the rest of the body. A single cilium is a cell’s eyes and nose, GPS receiver and even weather vane.
“If you lose your primary cilia, you’re pretty much unable to interact with your environment,” says Bradley Yoder, a cell and developmental biologist at the University of Alabama at Birmingham.
In the last decade, researchers have pegged a host of health issues on malfunctioning primary cilia. Snapped or otherwise inoperative appendages can lead to kidney failure, cancer, cleft palate, extra fingers or toes, water on the brain, hardened arteries, obesity, high blood pressure and heart disease.
More recently, teams have also started getting a handle on what exactly goes wrong to lead to primary cilia–related diseases, disorders and developmental defects, collectively known as ciliopathies. Primary cilia turn out to be the main receivers for messages sent by a well-studied, prolific protein that guides an embryo’s development and keeps cell growth in check.
But cilia are not like TV aerials that get only a few channels; crucial messages don’t have to begin with a chemical signal. Mechanical forces, such as blood, urine and bone pushing and pulling on cilia, can also get communication started.
As researchers learn more about how these cellular appendages function, the efforts could point to new therapies to bring disrupted receivers back online.
Getting attention
To say that primary cilia were completely ignored is not entirely fair. For a century, scientists have studied primary cilia in the eyes and nose. The rod and cone vision cells of the retina, in the back of the eye, stack light-gathering disks of proteins in their primary cilia. And primary cilia on specialized nerve cells in the nose, the only known exception to the one-per-cell rule, sense odors and react to them.
Such cilia, though, were viewed as anomalies. Similar singular protrusions from other cells were considered defunct leftovers with no purpose. Perhaps they were remnants of a murky past when cells swam solo, scientists thought (when they thought about primary cilia at all).
A first inkling that primary cilia other than those in the eyes and nose might have a purpose came from studies of people with polycystic kidney disease. This common genetic disorder strikes about one in a thousand people. It leads to large, fluid-filled cysts in the normally smooth kidneys that can result in organ failure and are a main reason for kidney dialysis.
Mutations in either of two genes cause a majority of polycystic kidney disease cases. When the genes were discovered in the 1990s, they were named for the disease: PKD1 and PKD2. But no one knew what role their protein products played. In 2002, Yoder and colleagues found the proteins in the primary cilia on kidney cells — strongly suggesting that the cilia play a part in proper kidney function.
In hindsight, researchers probably should have suspected that cilia defects are behind cystic kidneys, says Maxence Nachury, a cell biologist and biochemist at Stanford. Clues gleaned from experiments with mice pointed toward the cilia as problem areas. A strain of mutant mice with cyst-distorted kidneys was found in 1994 to carry defects in a gene now known as IFT88. Thanks to work in green algae, researchers knew IFT88’s protein was involved with transporting molecules in the moving type of cilia, called flagella. The “IFT” stands for intraflagellar transport, the system that shuttles molecular cargoes in all cilia types.
But people with polycystic kidney disease didn’t appear to have mutations in the human version of IFT88, so it wasn’t initially obvious that primary cilia would be to blame for the disease. Yoder’s work strongly pointed to cilia as the culprits.
Armed with just the knowledge that cilia defects lead to kidney cysts, scientists still didn’t fully appreciate what primary cilia could do, Nachury says. It took two more discoveries to cement cilia as a major force for human health and illness.
Cilia respected
Finding the cause of an obscure disease called Bardet-Biedl syndrome was the first big find. Patients with Bardet-Biedl syndrome, or BBS, experience a cluster of seemingly unrelated symptoms including obesity and vision loss, usually before the teen years. Many people with BBS have an extra pinky finger or toe. Some have heart defects. Cystic kidneys, learning disorders and loss of the sense of smell are some other common symptoms. Organs in a few people with the syndrome are even found on the wrong side of the body, though this may stem from a more general problem involving the moving kind of cilia (SN: 9/26/09, p. 26).
Today, these symptoms collectively scream “ciliopathy” to someone in the know. But not back in 2000 when the first BBS-linked genes were discovered. In 2003, proteins produced by these genes were found to crowd around the bases of primary cilia. Nachury and others have since discovered that seven BBS proteins and additional auxiliary proteins form a complex molecular machine known as the BBSome (pronounced bee-bee-sohm). That machine works like a bouncer sitting outside the passageway between the cilia and the rest of the cell, only taking down the velvet rope to allow select molecules into the club.
Discovering that disrupting the BBSome screws up cilia and leads to Bardet-Biedl syndrome gave primary cilia a bit of respect. But it still wasn’t clear how problems with the little hairs could lead to such a wide variety of disorders. An answer to that question came from studying a prolific development protein, called hedgehog.
Hedgehog was first discovered in fruit flies. It sets an organism’s body pattern from head to toe and right to left and carries out many other jobs in the body even after development is done, such as controlling when cells divide. Errors in hedgehog communication lead to birth defects, a type of skin cancer called basal cell carcinoma and perhaps heart disease. It’s such a big-deal molecule that mammals have three versions, the most famous called sonic hedgehog.
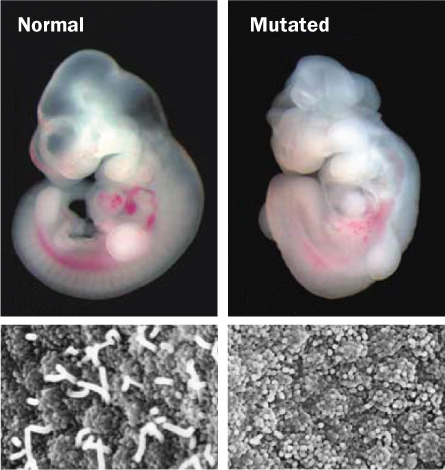
Around the same time as the BBS result, Kathryn Anderson of Memorial Sloan-Kettering Cancer Center in New York City and colleagues were looking for genetic mutations that could scramble sonic hedgehog signals in mice. The researchers found that the genes controlling molecular traffic within flagella and primary cilia must be intact for sonic hedgehog to get its message across. Disrupting those genes interrupts sonic hedgehog’s signal, so cells don’t know where they are in the body or what they should be when they grow up, Anderson and others established.
Finding out that primary cilia are hedgehog receivers was the second respect-delivering discovery, because it revealed that these cilia are involved in development, are needed throughout life and play a role pretty much everywhere in the body, Nachury says. It also explained why they could be behind so many disparate types of diseases and defects.
Now that cilia are getting some respect, efforts have turned to studying how they work.
Sonic hedgehog, it turns out, floats up to a cell and docks with another protein called Patched1 in a cilium’s outer membrane. This docking initiates a multistep biochemical reaction, sort of like knocking over the first in a long line of dominoes. As hedgehog bumps into Patched1, Patched1 releases Smoothened, which then topples the next domino and so on until the cascade activates genes that dictate what a cell will become or when it will divide.
Although cilia look as if they flow right into the rest of the cell, they are actually separate compartments with built-in barriers. Jeremy Reiter, a developmental biologist at the University of California, San Francisco, is trying to find out how to adjust the strength of these barriers. The efforts could lead to drugs for fighting some types of cancer, such as basal cell carcinoma.
In 2009, Reiter and his colleagues found that this cancer arises partly because Smoothened jumps the gun and starts knocking over other dominoes when it isn’t supposed to. More recently, Reiter and colleagues have found 12 different compounds that bar Smoothened from the cilia, one of which may help scientists identify additional protein players. The team reported the results August 21 in the Proceedings of the National Academy of Sciences.
Jobs pile up
While researchers explore primary cilia’s inner workings, others keep adding to the job list of these newly appreciated antennas.
For one, primary cilia on stomach cells help mice sense fat content in their diets and adjust stomach acid production, Juanita Merchant, a gastroenterologist at the University of Michigan, and colleagues reported in the August FASEB Journal. Mice fed a high-fat diet retracted primary cilia from stomach cells, the researchers found. Over time, this led to a drop in stomach acid levels. Less acidity could pave the way for stomach bacteria to move in and cause inflammation and possibly cancer, Merchant says.
But primary cilia sense more than chemical signals. They also pick up messages sent by mechanical forces, such as the flow of urine in the kidney. Kidney cells lining the tubes where urine flows dangle their cilia in the current. The force of the passing urine bends the cilia, triggering a different chain reaction from the one set off by hedgehog; this time one that starts with the proteins linked to polycystic kidney disease and ends with the cell electing not to multiply. Pulling dominoes from this chain means that kidney cells don’t get the message to sit tight, causing inappropriate cell growth that can produce the cysts seen in the disease.
Not only do people with mutated PKD genes have kidney cysts, many also have heart defects or high blood pressure. Beerend Hierck, a cell and developmental biologist at Leiden University Medical Center in the Netherlands, now blames broken antennas for contributing to these problems too.
Hierck didn’t set out to study primary cilia. He was, and still is, interested in heart development. While examining heart valve defects in chickens, Hierck noticed that some areas of the heart had bristly bits that made them rough, while others were smooth.
With painstaking electron microscopy work and other techniques, Hierck and his colleagues established that the bristles protruding from some heart cells are actually primary cilia. These cilia are involved in measuring how fast and in which direction blood is flowing.
This monitoring is particularly important during heart development. As the heart grows, blood flow gives clues to which cells should remain in the heart lining and which will migrate and form heart valves and other structures, the researchers reported last year in Circulation Research. The findings could help explain why children with absent or malfunctioning cilia sometimes are born with heart valve defects.
Skeletal defects have been linked to faulty mechanical sensors as well. About three years ago when David Hoey, a biomedical engineer at the University of Limerick in Ireland, started studying how bone cells sense mechanical forces, he’d never heard of primary cilia. “I didn’t really know what it was,” he says. “I had to Google it.”
Now, Hoey and others are building a case that primary cilia are stretch detectors that use the information they get to balance the constant building up and tearing down of bone.
Hoey studies osteocytes, octopus-like cells that hide in bones and reach their tentacles through crevices and tunnels to touch other types of cells responsible for the building and recycling of bone. Osteocytes need to measure mechanical forces to determine whether more bone is required or if the recyclers should be unleashed. Last year, Hoey and colleagues reported in Biochemical and Biophysical Research Communications that primary cilia on osteocytes sense fluid flow in bones.
It is unlikely that fluid flow bends the cilia of bone cells the way it does for cells in the kidney, he says. There’s just no room in the dense matrix where osteocytes sit. Instead, the cilia probably detect stretching of the matrix occurring because of that fluid flow, sort of like a spider feeling its web wiggle. Figuring out how primary cilia tap into this spidey sense may point to possible treatments for conditions such as osteoporosis.
While most researchers are concentrating on understanding how primary cilia receive and interpret chemical, mechanical and other messages, Joel Rosenbaum, a cell biologist at Yale University, is thinking about the cilia in a new way. Rosenbaum’s lab has long studied the flagella of the single-celled algae Chlamydomonas, and made major contributions to the understanding of the intraflagellar transport system. Now, he is exploring the notion that primary cilia might be transmitters as well as receivers.
A bit of evidence suggests he could be on to something. In 2009, a Japanese group showed that young Chlamydomonas release an enzyme called sporangin from their flagella during hatching. The enzyme triggers the tearing down of the mother cell’s walls. Rosenbaum thinks the algae may send other signals via their flagella too.
If so, human cells may use their primary cilia as short wave communication systems, and transmission errors could be behind a whole new list of diseases. Rosenbaum is amassing preliminary data to support the idea, but he can’t discuss the results until they are published.
Stay tuned. It’s a good bet your primary cilia will be.
Pack ’em in
Source: M.R. Mahjoub and T. Stearns/
Current Biology
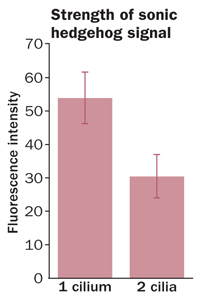
Although scientists now recognize the importance of primary cilia, it is still a mystery why cells create the compartments in the first place. Wouldn’t it be better to place listening stations around the entire cell, to catch whispers coming from different quarters? One leading explanation is that cramming all of a cell’s receiving equipment into one radio closet might make it easier for message-relaying proteins to find each other, thus strengthening the signal.
Moe Mahjoub and Tim Stearns of Stanford University recently delivered some data to support the idea by creating cells bearing more than one primary cilium. Doubling or tripling the number of cilia on a cell meant that messages sent by an important development protein called sonic hedgehog were diluted by the time they reached their targets, the researchers reported in the Sept. 11 Current Biology.







