Out on a Limb
The science of body development may make kindling out of evolutionary trees

Over the past 25 years, paleoanthropologists have nurtured one evolutionary tree after another, hoping to reap ever sturdier portrayals of humanity’s descent. Each of these trees sprouts out of an approximately 5-million-year-old presumed common ancestor and then branches along lines for various australopithecine and Homo species, ending at Homo sapiens. Recently, investigators have become fond of assigning new fossil finds to unique rather than established species, so evolutionary trees have gotten downright bushy.
When paleoanthropologists craft evolutionary trees, they usually select bone and tooth measurements, sometimes as many as several hundred from different fossils, to plug into a computerized statistical analysis. This technique, called cladistics, places species on related limbs of the tree according to the number of recently evolved skeletal characteristics that they share with each other but with no other species. Scientists also have applied cladistics to differences in the nucleotide sequence of segments of DNA.
However, a growing number of investigators, including some formerly ardent evolutionary-tree nurturers now suspect that the branching cladistic creations suffer from conceptual root rot. The whole enterprise rests on shaky biological and misleading statistical assumptions, they say.
Anthropologists first need to enter the unfamiliar territory of developmental biology and learn how complex chains of molecular processes form the bones and soft tissues that coalesce into bodies, argues Tim D. White of the University of California, Berkeley.
As a codiscoverer more than 25 years ago of the famous 3.2-million-year-old remains of the human ancestor now known as Lucy, White has spawned his share of evolutionary trees. Over the past decade, though, advances in developmental biology have led him to abstain from that practice.
Biological research now suggests that as an organism grows from fetus to adult, the shape and arrangement of its bones reflect a cascade of molecular processes that were ignited by certain genes but then responded flexibly to a variety of influences in their biological milieu. So, genuinely informative skeletal traits for evolutionary trees are relatively uncommon and can’t be identified by simply eyeballing specimens, he says.
“You can’t [break up] skeletal anatomy, put all of the traits into a [statistical] program, and generate something that makes biological sense,” White asserts. “A lot of people who have published cladistic trees are going to be in trouble.”
Popular tools
White may be right, but statistical comparisons of our fossil ancestors’ skeletal features are likely to remain popular, remarks anthropologist Erik Trinkaus of Washington University in St. Louis. Many researchers prefer to explore evolution with cladistic analyses of bones, DNA, or both, he says.
Agreement on the general technique doesn’t imply agreement on the best data to use with it. Scientists frequently disagree about which skeletal traits are recently evolved and which specimens share them.
Skull and tooth measurements from people, chimps, gorillas, and orangutans—the same measurements frequently employed to generate evolutionary trees for human ancestors—give rise to a primate evolutionary tree that clashes with the genetic version, reported Bernard Wood of George Washington University in Washington, D.C., and Mark Collard of University College, London, in the April 25 Proceedings of the National Academy of Sciences. These genetic data have set an evolutionary standard that skeletal traits can’t match, Wood and Collard concluded.
Genetic studies have converged on the view that people bear a particularly close evolutionary relationship to chimpanzees, while gorillas and orangutans arose along separate evolutionary lines, according to these researchers. However, Wood and Collard’s cladistic analysis of skeletal traits places chimps, gorillas, and orangutans along closely related evolutionary branches, with people on a more distant limb.
A separate, broader cladistic analysis, which includes a comparative set of skeletal measurements from baboons and several other African monkeys as well as the apes, places humans and orangutans in one evolutionary group and gorillas and chimpanzees in another.
However, 197 measurements of muscles, nerves, blood vessels, and connective tissue from various modern primates generate an evolutionary picture that corresponds to the established DNA version, according to Wood, Collard, and Sally Gibbs of George Washington University.
The researchers’ cladistic analysis of soft-tissue features, such as the branching patterns of specific arteries and the number of fingers that receive input from certain nerves to the hand, appears in the Sept. 26 Proceedings of the National Academy of Sciences.
Muscle attachments to bone and other soft-tissue traits may remain far more stable than bone itself does during individual development, comments anthropologist David Pilbeam of Harvard University. Perhaps some of the marks left on bones by muscle attachments and blood vessels may hold critical evolutionary clues for fossil investigators, Wood suggests. He and his colleagues are now looking for such cladistic candidates.
Cladistics produces valid evolutionary insights only when it’s used to analyze numerous parts of an organism that have developed independently of one another. For instance, evolutionary primate-tree builders typically assume that different sets of genes orchestrate the shapes of the jaw and of the forehead, rendering measurements of both features suitable for cladistic analysis.
Ongoing research in developmental biology raises ominous warning signs about such assumptions. It indicates that many features of bones and teeth arise as by-products of other traits during an individual’s skeletal development.
Wood, who until recently was an avid and influential evolutionary tree builder, now doubts the accuracy of cladistics as currently practiced. “I’m eating large bowls of humble pie these days,” Wood says. “My defense is, ‘If you make a mess, you need to clean it up.'”
Evoking controversy
Genetic approaches to evolutionary-tree building also evoke controversy (SN: 2/6/99, p. 88: http://www.sciencenews.org/pages/sn_arc99/2_6_99/bob1.htm). For instance, one scientific camp holds that Neandertals’ DNA composition pegs them as a dead-end species (SN: 7/19/97, p. 37). Trinkaus and others view the genetic data as ambiguous and say that a recent fossil find exhibits a mix of traits produced by interbreeding of Neandertals and modern humans (SN: 5/8/99, p. 295: http://www.sciencenews.org/pages/sn_arc99/5_8_99/fob7.htm).
Such debates will prove fruitless, not to mention endless, unless scientists manage to discern how bones develop and which of their features reflect direct genetic effects, White says. Along with anthropologist C. Owen Lovejoy of Kent (Ohio) State University and biologist Martin J. Cohn of the University of Reading in England, White coauthored a paper in the Nov. 9, 1999 Proceedings of the National Academy of Sciences summarizing what’s known about the biology of bone development.
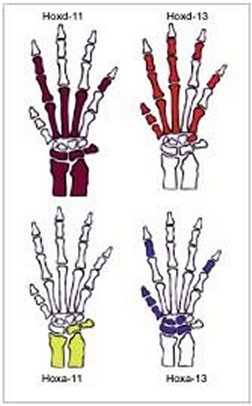
Lovejoy’s group devotes particular attention to vertebrate DNA sequences known as homeobox genes, which biologists have studied over the past 15 years. Homeobox genes control the general shape of the fetal body. These genes also trigger a series of biological processes that foster the growth of integrated sections of the skeleton rather than the development of individual bones, Lovejoy asserts.
For instance, the same two homeobox genes appear to regulate the growth of both the vertebrate forearm and all fingers except the thumb, whereas a couple other of these genes participate only in thumb growth, Lovejoy and his colleagues report in an upcoming Evolution and Development. Relatively subtle alterations of these genes or their protein products at various times could easily have accounted for the evolution of hand proportions typical of modern primates and of our fossil ancestors, the researchers theorize.
Lovejoy and his colleagues used chemical probes injected into mouse embryos to establish that one pair of homeobox genes displays a distinctive molecular footprint in the thumb bones, while another pair leaves a unique calling card in bones of the forearm and the remaining four fingers.
In a sample of 65 skeletons from people, apes, and monkeys, Lovejoy’s team then found that the adult forearm is longer or shorter in concert with corresponding size differences in all fingers except the thumb. In contrast, from one skeleton to the next, the presence of a short or long thumb showed no relationship to a short or long forearm or the lengths of the other fingers.
Evolutionary tweaking of the two pairs of homeobox genes may have yielded hand proportions ranging from the sturdy thumbs attached to short forearms and fingers typical of modern humans, to the tiny thumbs affixed to elongated forearms and fingers characteristic of some monkeys, Lovejoy says.
Fossil evidence from human ancestors indicates that marked growth of the thumb and reduction of the other fingers occurred between 2.5 million and 1.5 million years ago, a time that featured the emergence of Homo, the line of direct human ancestors. Homeobox-gene modifications probably triggered this evolutionary shift to hands capable of making and using stone tools, Lovejoy holds.
A similar scenario may explain tooth evolution in human ancestors and have implications for much of the rest of the skull, according to anatomist Melanie A. McCollum of Case Western Reserve University in Cleveland and biologist Paul T. Sharpe of Kings College London.
Recent experiments on tooth development in mice indicate that separate homeobox genes regulate the growth of teeth in the upper and lower jaws and at the front and back of the mouth, McCollum and Sharpe report in an upcoming BioEssays.
Alterations of these genes may have spurred the relatively rapid evolution around 2.5 million years ago of so-called robust australopithecines, who had smaller brains than their predecessors and greatly expanded chewing teeth at the back of the mouth, McCollum says. She’s already proposed that the unusual tooth proportions of robust australopithecines prompted the formation of many other of their distinctive skull features (SN: 4/24/99, p. 267).
“In early human ancestors, evolutionary forces seem to have operated on genes that affected the teeth but not much else in the face,” McCollum asserts. “Yet in most cladistic analyses, dental traits seem to play a minor role compared with skull measurements.”
Quick change
Alterations to homeobox genes appear capable of quickly reshaping the body’s layout and generating new species without having to progress through a series of transitional forms, says anthropologist Jeffrey H. Schwartz of the University of Pittsburgh in his book Sudden Origins (1999, Wiley).
“There’s a tremendous amount to be done before the key molecular pathways for [body] development are worked out,” Schwartz says.
In an attempt to spur research into the genetics and biology of body development, anthropologist Timothy G. Bromage of City University of New York’s Hunter College has submitted a proposal to the National Science Foundation to establish a Science and Technology Center for Comparative Bone and Skeletal Structure. The 10-year, $30-million venture would include 34 scientists—including Lovejoy and McCollum—from 20 public and private institutions.
Today, many anthropologists show little interest in how biological development shaped the bodies of our fossil ancestors, Bromage says. In contrast, medical researchers have launched a number of projects to unravel processes of bone formation and repair involved in diseases such as osteoporosis (SN: 1/15/00, p. 41: Boning Up). That work, along with findings in genetics and developmental biology, would inspire research at the proposed bone-and-skeletal-structure center, according to Bromage.
“We can’t solve debates over Neandertals or any other human ancestors using anatomical characteristics that are subjectively defined and don’t have a clear relationship to evolutionary history,” he remarks. “Let’s examine bones with a fresh perspective on biology and life history.”