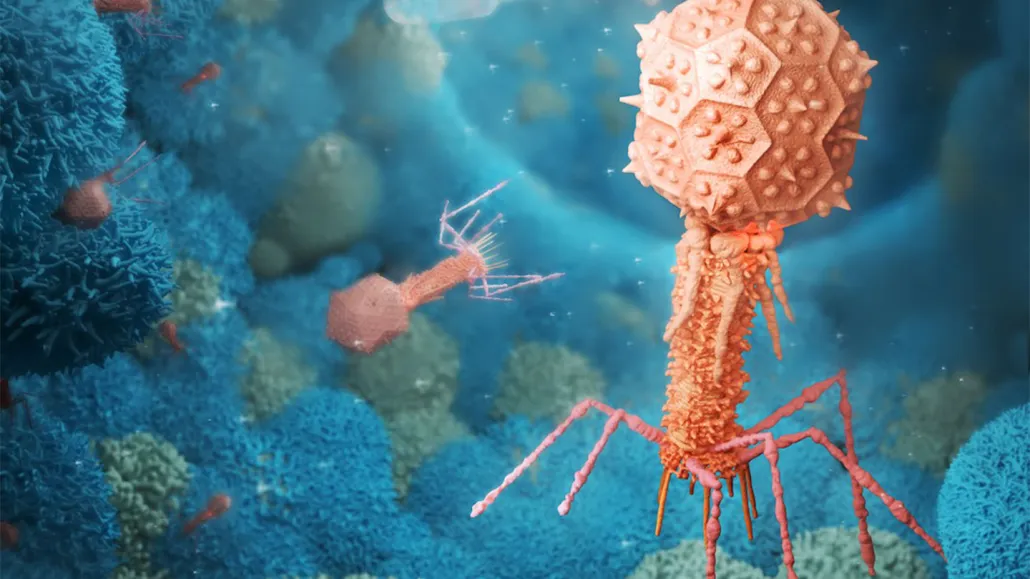
Bacteriophages were long thought to only interact with the bacteria they infect. But recent research has shown they can be ingested by human cells, and now there is evidence that our cells can use them as a food source. This illustration shows bacteriophages interacting with mammalian cells.
T2Q and Barr Lab (CC-BY 4.0)







