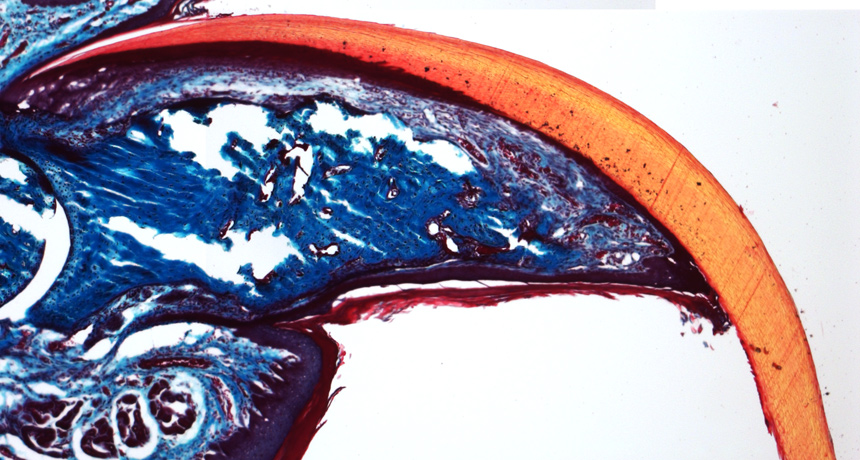
Takeo et al/Nature 2013
Clumsy manicurists can thank a set of stem cells under the base of the fingernail for erasing their mistakes. Those cells allow not only trimmed fingernails but amputated fingertips to regrow. Doctors could one day use nail stem cells to treat malformed nails or even amputated limbs, Mayumi Ito of New York University Langone Medical Center suggests.
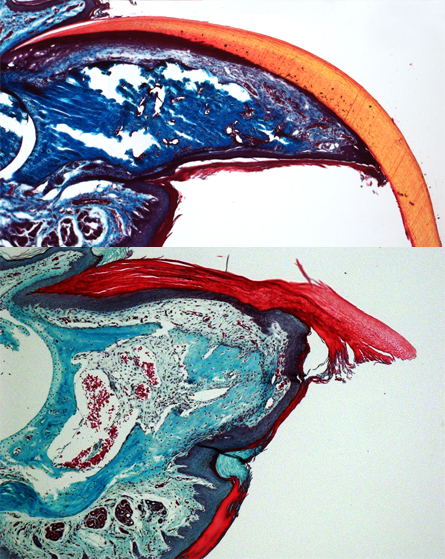
Scientists have long known that children and some adults can regrow the tips of their fingers after amputation. But digits can’t regenerate if more than the nail region is amputated.
Ito and her colleagues traced the fates of cells on the back feet of mice during nail growth and found a population of stem cells that produces the hard part of the nail and the soft tissue underneath. When the researchers cut off the end of a toe, signals from the regrowing nail stimulated the tissue underneath to form new bone, the authors report June 12 in Nature.
The digit bones can regenerate only if the amputated stump still has some nail stem cells, the researchers found. But the cells alone are not enough; also crucial is a zone of tissue that grows from the stem cells during normal nail growth. After amputation, this tissue sends signals that attract new nerves into the end of the stump and begin the bone regeneration process. If amputation removes the nail zone or if the signals are blocked, the digits will not regenerate.
When the researchers genetically manipulated the mice to turn on the regeneration signals, nail stem cells alone could spur digit regeneration even without the neighboring nail tissue zone.
Other researchers have found that similar signals are involved in regenerating amputated amphibian limbs. “We were really amazed by the similarity between these processes,” Ito says. The parallels suggest that mammals might retain some of the newt’s famous power to regrow entire legs.
The similarity between mammalian and amphibian regeneration is encouraging, says Ken Muneoka of Tulane University. That parallel, he says, “gives us hope that we will be able to induce human regeneration in the not too distant future.”







