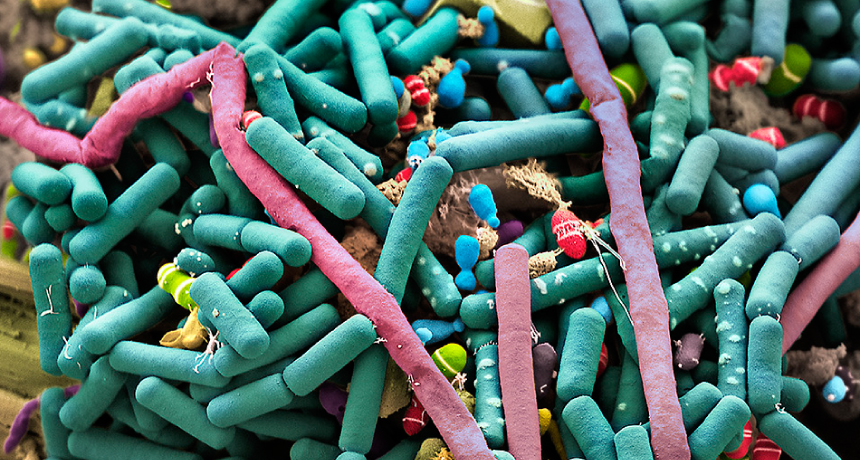
MICROBE MECCA About a thousand species of bacteria reside in the human gut, some of which are displayed in this hand-colored scanning electron micrograph.
Martin Oeggerli
1
We are not alone. Humans’ vast inner and outer spaces teem with a menagerie of microbes that stand poised to alter conceptions of what and who we are.
Traditionally, microbes have been viewed as insidious invaders that make people sick or as freeloaders in the human gut. That view is beginning to change. In 2013, scientists amassed substantial evidence that people and other animals form a unit with their resident bacteria, archaea, fungi and viruses — the collection of microbes known as the microbiome. In fact, only about 10 percent of a person’s cells are human; microbes make up the other 90 percent.
Many researchers point out that ultimately, every species is out for itself. Nevertheless several new studies argue in favor of considering animals as superorganisms composed of host and microbes. Some scientists even advocate lumping a host organism’s genes with those of its microbes into one “hologenome.”
Treating a host, such as the human body, and its resident bacteria as a unit — or at least as an ecosystem with intimately interconnected parts — offers various benefits, scientists say. The superorganism approach may help researchers better understand how diet, chemicals and other environmental factors affect health, for instance.
Everyone, including identical twins, carries a slightly different microbial mix. Strong evidence indicates that some differences stem from diet or habitat. But even mice raised under uniform lab conditions still have individualized microbiomes. In October, two groups presented research suggesting that host genes play a role in selecting which microbes are allowed to settle in and on the body (SN: 11/30/13, p. 11). Immune system genes may be especially important in screening suitable microbial companions.
People with immune system problems have more types of bacteria and fungi on their skin. New research shows that some of those microbes may contribute to eczema-like rashes. That finding supports the idea that the immune system grants visas to friendly microbes while keeping out dangerous interlopers.
Newborns rein in their own immune systems to allow bacteria to take hold, one study found (SN: 12/14/13, p. 10). Previously, researchers thought that babies’ immune systems were just too immature to control microbes. But the new work shows that in mice and human umbilical cords, blood cells carry an immune-suppressing protein that prevents defenders from fighting off beneficial bacteria.
In mice, pups of stressed moms picked up a different mix of bacteria during birth than those born tonon-stressed moms, researchers reported at the Society for Neuroscience meeting in November. Those bacteria may affect early brain development and possibly contribute to disorders such as autism and schizophrenia (SN: 12/14/13, p. 13).
A study reported in December may strengthen the link between autism and gut microbes ( SN Online: 12/5/13 ). Caltech researchers found that mice with autism-like symptoms have a different mix of gut microbes than normal mice do. Those microbes make chemicals that leak from the intestines into the bloodstream (and perhaps the brain), producing behavioral changes. Treating the mice with the beneficial bacterium Bacteroides fragilis improved some symptoms, suggesting that altering the microbial mix might help some children with autism.Once established, friendly bacteria shield their hosts from harmful invaders and may keep the immune system from overreacting. Harvard researchers discovered that some intestinal microbes make immune-calming molecules that can help reduce the kind of inflammation that afflicts the bowels in diseases like colitis (SN: 8/10/13, p. 14).
Even friendly bacteria put their own needs first, though. Another Harvard group found that some strains of a common gut microbe called Eggerthella lenta can rob heart patients of a drug called digoxin if the bacteria don’t get enough protein from their hosts (SN Online: 7/19/13). Some microbes change chemicals in meat into artery-cloggers (SN: 5/18/13, p. 14) or cause pain all on their own (SN: 10/5/13, p. 16).
Microbiomes not only alter the biochemical milieu in individuals, but can also influence relationships between entire species. Or even the course of evolution. A study of jewel wasps, for instance, suggests that their microbiomes can prevent two species from successfully breeding with one another (SN: 8/10/13, p. 13).
Hybrid male offspring of the two species die as larvae, an effect long explained as incompatibility between the species’ genes. But when Seth Bordenstein of Vanderbilt University and his colleague Robert Brucker removed microbes from the hybrid larvae, the wasps survived. That finding indicates that microbes in the wasps’ guts and not just the wasp genes contribute to keeping the two species from interbreeding.
The microbial momentum continues to build. Ongoing research is sure to find other ways in which microbes and their hosts interact, for good and ill. “It’s not just a one-way street,” says dermatologist Heidi Kong of the National Institutes of Health in Bethesda, Md. “The microbes are doing something to us and we are doing things to our microbes.”







